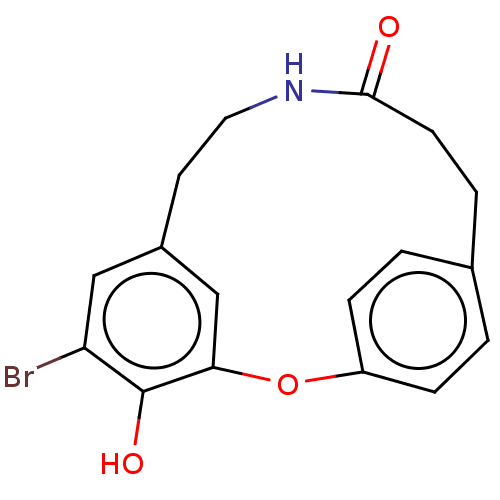
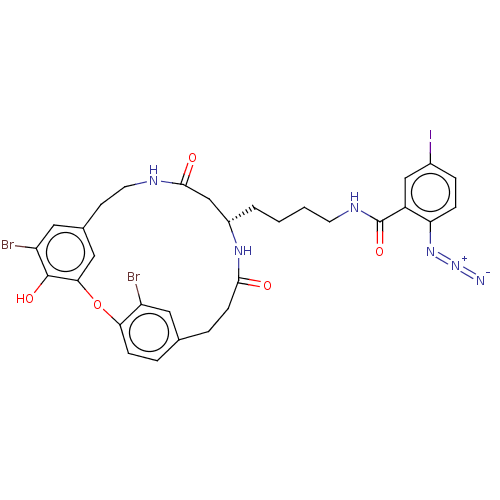
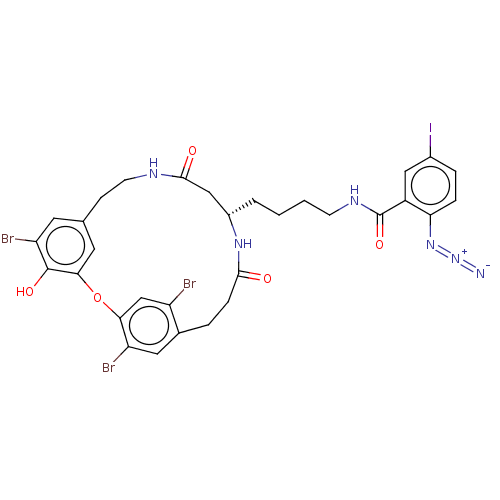
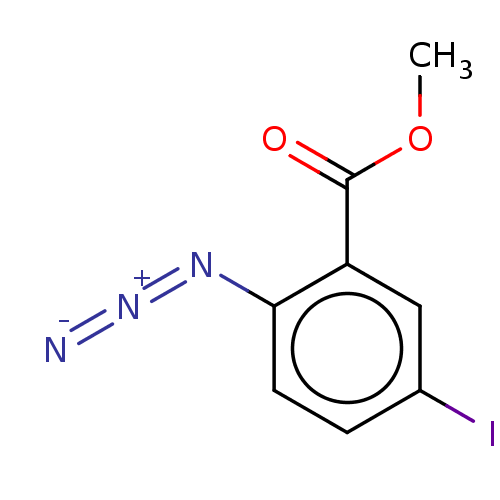
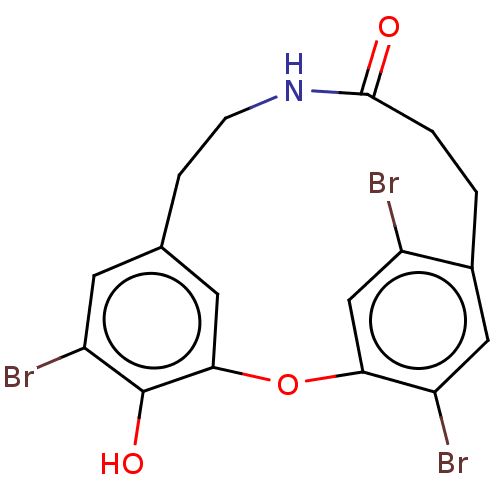
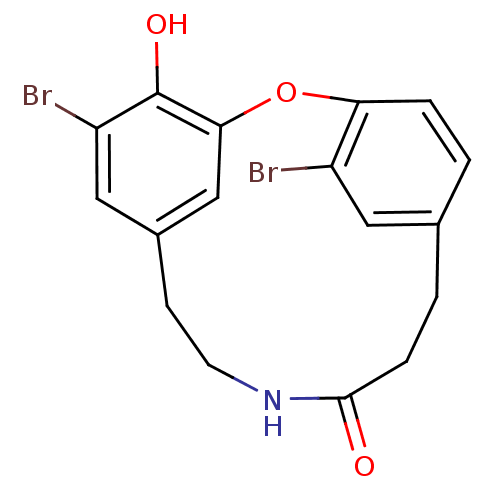
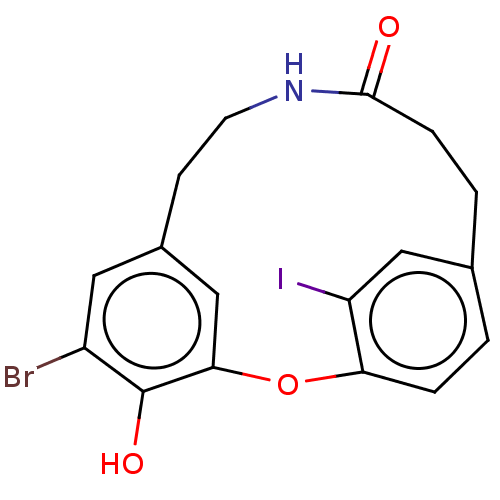
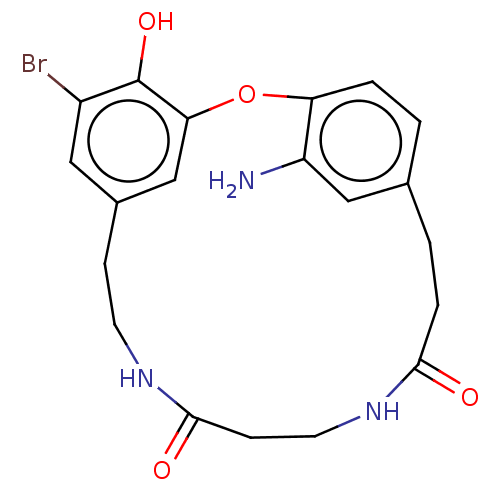
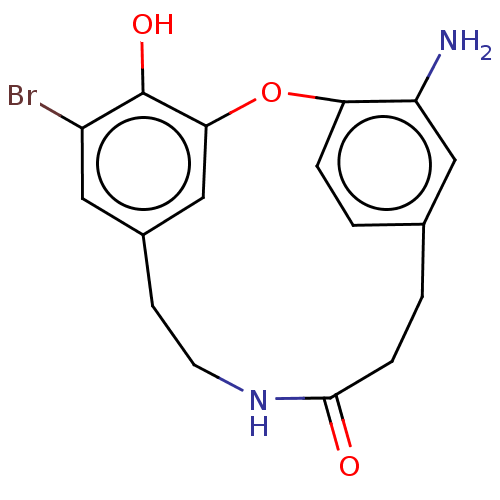
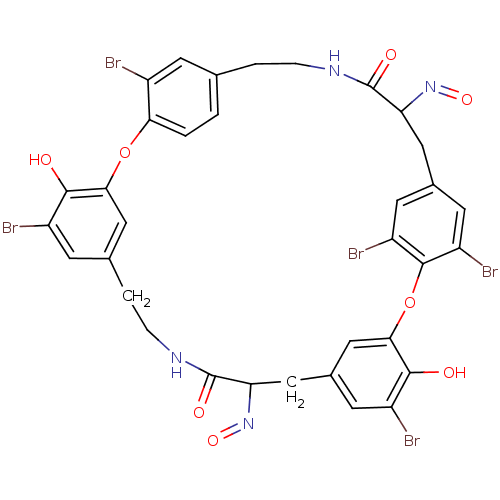
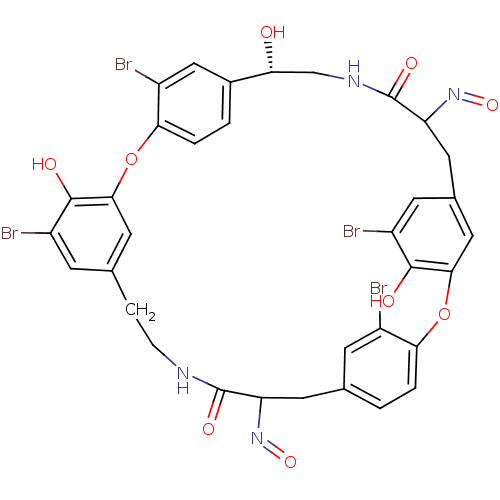
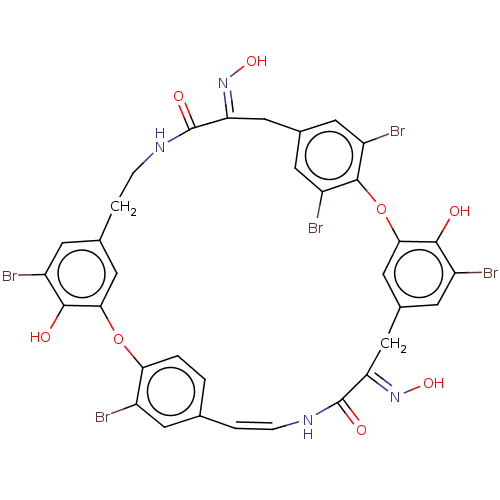
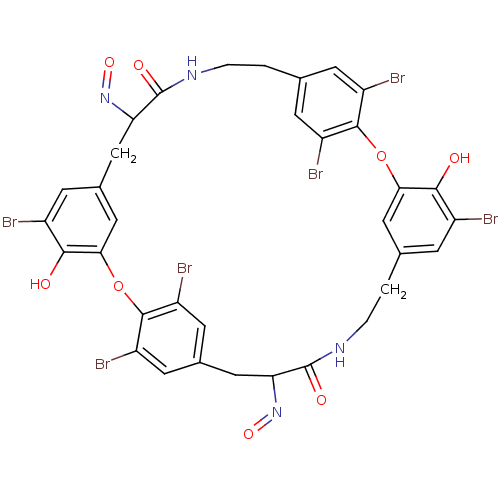
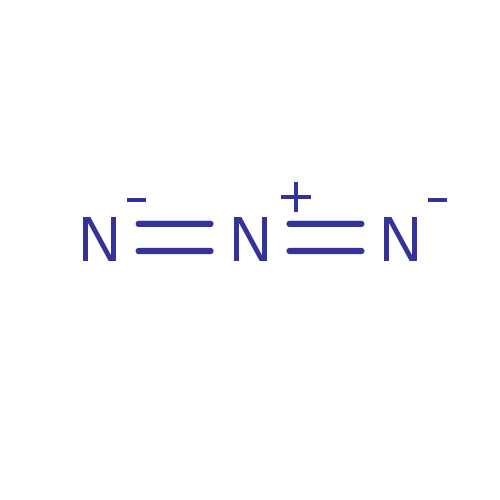Affinity DataIC50: 0.300nMAssay Description:Inhibition of cathepsin EMore data for this Ligand-Target Pair
Affinity DataIC50: 4.70E+3nMAssay Description:Inhibition of Trametes versicolor laccase assessed as production of dopachrome using 100 uM epinephrine as substrate preincubated for 30 mins before ...More data for this Ligand-Target Pair
Affinity DataIC50: 4.90E+3nMAssay Description:Inhibition of Trametes versicolor laccase assessed as production of dopachrome using 200 uM dopamine as substrate preincubated for 30 mins before sub...More data for this Ligand-Target Pair
Affinity DataIC50: 5.80E+3nMAssay Description:Inhibition of Trametes versicolor laccase assessed as production of dopachrome using 300 uM L-dopa as substrate preincubated for 30 mins before subst...More data for this Ligand-Target Pair
Affinity DataIC50: 2.57E+5nMAssay Description:Inhibition of Trametes versicolor laccase assessed as production of dopachrome using 200 uM dopamine as substrate preincubated for 30 mins before sub...More data for this Ligand-Target Pair
Affinity DataIC50: 2.63E+5nMAssay Description:Inhibition of Trametes versicolor laccase assessed as production of dopachrome using 100 uM epinephrine as substrate preincubated for 30 mins before ...More data for this Ligand-Target Pair
Affinity DataIC50: 2.79E+5nMAssay Description:Inhibition of Trametes versicolor laccase assessed as production of dopachrome using 300 uM L-dopa as substrate preincubated for 30 mins before subst...More data for this Ligand-Target Pair
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A/Ryanodine receptor 1(Homo sapiens (Human))
University of California
Curated by ChEMBL
University of California
Curated by ChEMBL
Affinity DataEC50: 2.40E+4nMAssay Description:Agonist like activity at skeletal junction sarcoplasmic reticulum RyR1/FKBP12 complex assessed as increase in [3H]ryanodine bindingMore data for this Ligand-Target Pair
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A/Ryanodine receptor 1(Homo sapiens (Human))
University of California
Curated by ChEMBL
University of California
Curated by ChEMBL
Affinity DataEC50: 2.00E+4nMAssay Description:Agonist like activity at skeletal junction sarcoplasmic reticulum RyR1/FKBP12 complex assessed as increase in [3H]ryanodine bindingMore data for this Ligand-Target Pair
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A/Ryanodine receptor 1(Homo sapiens (Human))
University of California
Curated by ChEMBL
University of California
Curated by ChEMBL
Affinity DataEC50: >1.00E+5nMAssay Description:Agonist like activity at skeletal junction sarcoplasmic reticulum RyR1/FKBP12 complex assessed as increase in [3H]ryanodine bindingMore data for this Ligand-Target Pair
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A/Ryanodine receptor 1(Homo sapiens (Human))
University of California
Curated by ChEMBL
University of California
Curated by ChEMBL
Affinity DataEC50: >1.00E+5nMAssay Description:Agonist like activity at skeletal junction sarcoplasmic reticulum RyR1/FKBP12 complex assessed as increase in [3H]ryanodine bindingMore data for this Ligand-Target Pair
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A/Ryanodine receptor 1(Homo sapiens (Human))
University of California
Curated by ChEMBL
University of California
Curated by ChEMBL
Affinity DataEC50: 6.50E+3nMAssay Description:Agonist like activity at skeletal junction sarcoplasmic reticulum RyR1/FKBP12 complex assessed as increase in [3H]ryanodine bindingMore data for this Ligand-Target Pair
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A/Ryanodine receptor 1(Homo sapiens (Human))
University of California
Curated by ChEMBL
University of California
Curated by ChEMBL
Affinity DataEC50: 2.20E+3nMAssay Description:Agonist like activity at skeletal junction sarcoplasmic reticulum RyR1/FKBP12 complex assessed as increase in [3H]ryanodine bindingMore data for this Ligand-Target Pair
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A/Ryanodine receptor 1(Homo sapiens (Human))
University of California
Curated by ChEMBL
University of California
Curated by ChEMBL
Affinity DataEC50: >1.00E+5nMAssay Description:Agonist like activity at skeletal junction sarcoplasmic reticulum RyR1/FKBP12 complex assessed as increase in [3H]ryanodine bindingMore data for this Ligand-Target Pair
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A/Ryanodine receptor 1(Homo sapiens (Human))
University of California
Curated by ChEMBL
University of California
Curated by ChEMBL
Affinity DataEC50: >1.00E+5nMAssay Description:Agonist like activity at skeletal junction sarcoplasmic reticulum RyR1/FKBP12 complex assessed as increase in [3H]ryanodine bindingMore data for this Ligand-Target Pair
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A/Ryanodine receptor 1(Homo sapiens (Human))
University of California
Curated by ChEMBL
University of California
Curated by ChEMBL
Affinity DataEC50: 2.09E+4nMAssay Description:Agonist like activity at skeletal junction sarcoplasmic reticulum RyR1/FKBP12 complex assessed as increase in [3H]ryanodine bindingMore data for this Ligand-Target Pair
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A/Ryanodine receptor 1(Homo sapiens (Human))
University of California
Curated by ChEMBL
University of California
Curated by ChEMBL
Affinity DataEC50: 1.09E+4nMAssay Description:Agonist like activity at skeletal junction sarcoplasmic reticulum RyR1/FKBP12 complex assessed as increase in [3H]ryanodine bindingMore data for this Ligand-Target Pair
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A/Ryanodine receptor 1(Homo sapiens (Human))
University of California
Curated by ChEMBL
University of California
Curated by ChEMBL
Affinity DataEC50: 2.86E+4nMAssay Description:Agonist like activity at skeletal junction sarcoplasmic reticulum RyR1/FKBP12 complex assessed as increase in [3H]ryanodine bindingMore data for this Ligand-Target Pair
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A/Ryanodine receptor 1(Homo sapiens (Human))
University of California
Curated by ChEMBL
University of California
Curated by ChEMBL
Affinity DataEC50: 2.10E+4nMAssay Description:Agonist like activity at skeletal junction sarcoplasmic reticulum RyR1/FKBP12 complex assessed as increase in [3H]ryanodine bindingMore data for this Ligand-Target Pair
TargetPeptidyl-prolyl cis-trans isomerase FKBP1A/Ryanodine receptor 1(Homo sapiens (Human))
University of California
Curated by ChEMBL
University of California
Curated by ChEMBL
Affinity DataEC50: 3.30E+4nMAssay Description:Agonist like activity at skeletal junction sarcoplasmic reticulum RyR1/FKBP12 complex assessed as increase in [3H]ryanodine bindingMore data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+5nMAssay Description:Displacement of [3H]ryanodine from Ry1R/FKBP12 receptor complex in rabbit sarcoplasmic reticulumMore data for this Ligand-Target Pair
Affinity DataEC50: 2.06E+4nMAssay Description:Displacement of [3H]ryanodine from Ry1R/FKBP12 receptor complex in rabbit sarcoplasmic reticulumMore data for this Ligand-Target Pair
Affinity DataEC50: 1.36E+4nMAssay Description:Displacement of [3H]ryanodine from Ry1R/FKBP12 receptor complex in rabbit sarcoplasmic reticulumMore data for this Ligand-Target Pair
Affinity DataEC50: 1.00E+5nMAssay Description:Displacement of [3H]ryanodine from Ry1R/FKBP12 receptor complex in rabbit sarcoplasmic reticulumMore data for this Ligand-Target Pair
Affinity DataEC50: 2.00E+3nMAssay Description:Displacement of [3H]ryanodine from Ry1R/FKBP12 receptor complex in rabbit sarcoplasmic reticulumMore data for this Ligand-Target Pair
Affinity DataEC50: >1.00E+5nMAssay Description:Displacement of [3H]ryanodine from RyR1 calcium channel in sarcoplasmic reticulum assessed as calcium mobilizationMore data for this Ligand-Target Pair
Affinity DataEC50: 5.80E+3nMAssay Description:Displacement of [3H]ryanodine from RyR1 calcium channel in sarcoplasmic reticulum assessed as calcium mobilizationMore data for this Ligand-Target Pair
Affinity DataEC50: 1.47E+4nMAssay Description:Displacement of [3H]ryanodine from RyR1 calcium channel in sarcoplasmic reticulum assessed as calcium mobilizationMore data for this Ligand-Target Pair
Affinity DataEC50: 2.60E+3nMAssay Description:Displacement of [3H]ryanodine from RyR1 calcium channel in sarcoplasmic reticulum assessed as calcium mobilizationMore data for this Ligand-Target Pair