Activity Spreadsheet -- Enzyme Inhibition Constant Data from BindingDB (change energy unit to kcal/mol)
 Found 57 hits in this display
Found 57 hits in this display
 BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
 BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
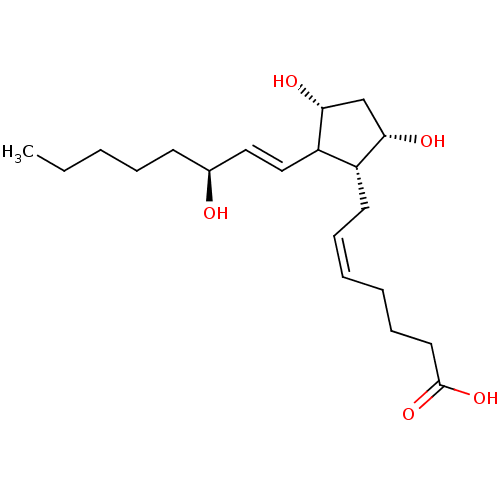
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
 BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
 BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
 BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
 BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
 BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
 BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
 BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
 BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
 BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
 BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
 BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
 BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
 BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
 BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
BDBM50020300((S-isomer)7-[3,5-Dihydroxy-2-(3-hydroxy-oct-1-enyl...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChIIC50: 43nMAssay Description:Binding potency towards Prostaglandin F2 alpha receptor (competitive binding) with natural [3H]-PGF 2 alpha in ovine luteal cells (OLC)More data for this Ligand-Target Pair
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChIEC50: 600nMAssay Description:Functional activity in RAT-1cells, transiently-transfected with human Prostaglandin E receptor EP1 (% of control ligand, 17-phi-PGE2=80%)More data for this Ligand-Target Pair
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChIEC50: 79nMAssay Description:Functional activity in RAT-1cells, transiently-transfected with human FP-receptor (% of control ligand, fluprostenol=100%)More data for this Ligand-Target Pair
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChIEC50: 2.30E+3nMAssay Description:Functional activity in RAT-1cells, transiently-transfected with human Prostaglandin E receptor EP3 (% of control ligand, sulprostone=50%)More data for this Ligand-Target Pair
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChIEC50: 1.40E+3nMAssay Description:Functional activity in RAT-1cells, transiently-transfected with human TP-receptor (% of control ligand, [3H]-U-46,619=95%)More data for this Ligand-Target Pair
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
 BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChI
BDBM50035622((5Z,13E,15S)-9alpha,11alpha,15-trihydroxyprosta-5,...)copy SMILEScopy InChIEC50: 24.5nMAssay Description:Efficacy for stimulation of prostanoid FP receptor-linked phosphoinositide turnover in Swiss 3T3 mouse fibroblast cellsMore data for this Ligand-Target Pair
Displayed 1 to 50 (of 57 total ) | Next | Last >>