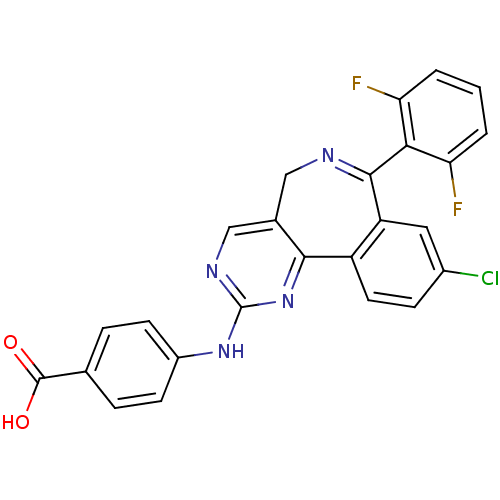
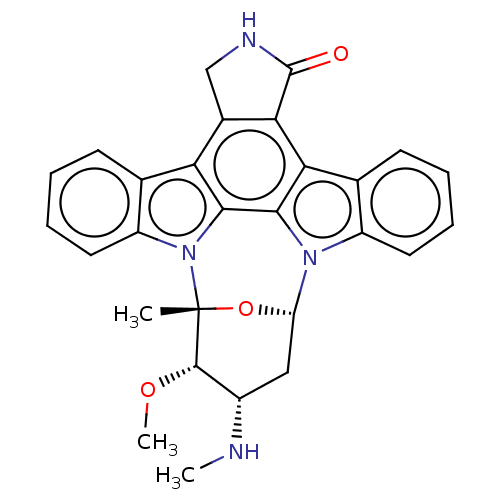
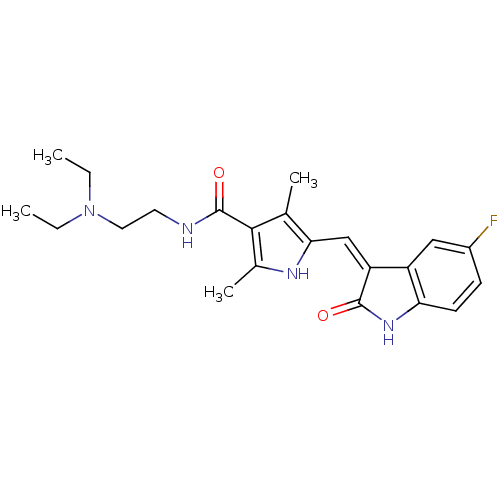
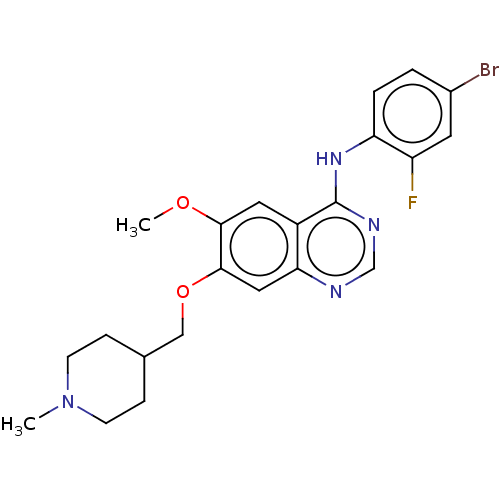
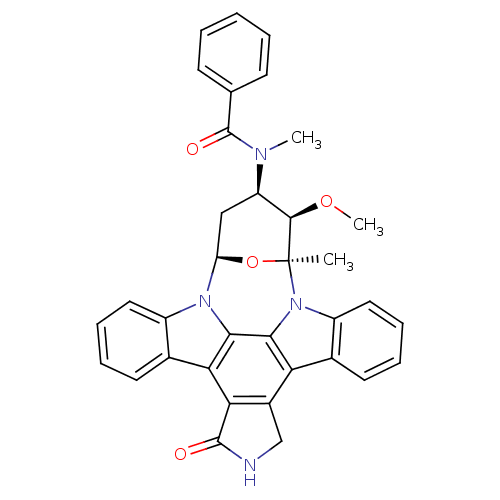
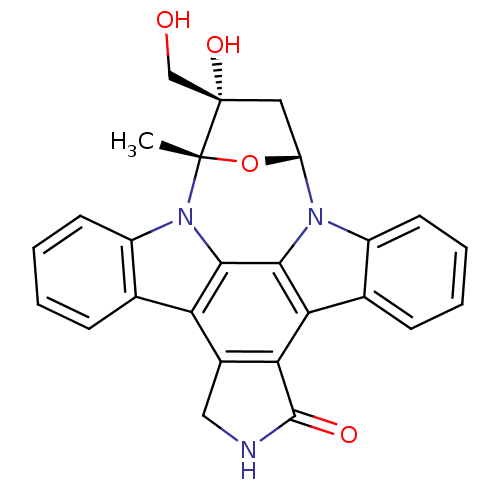
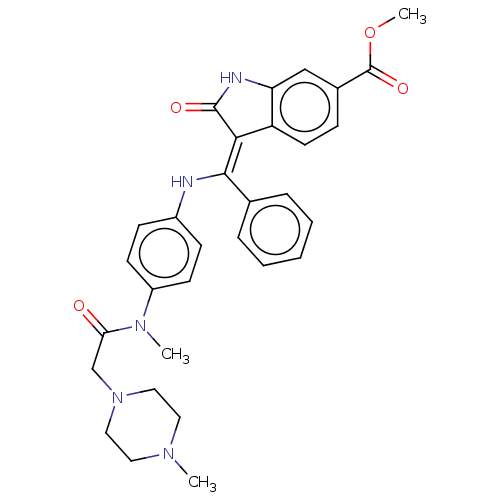
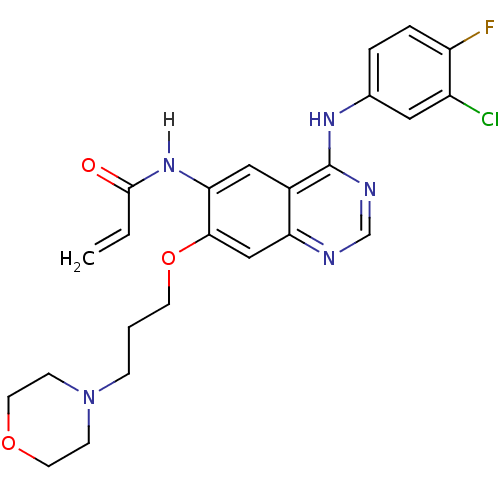
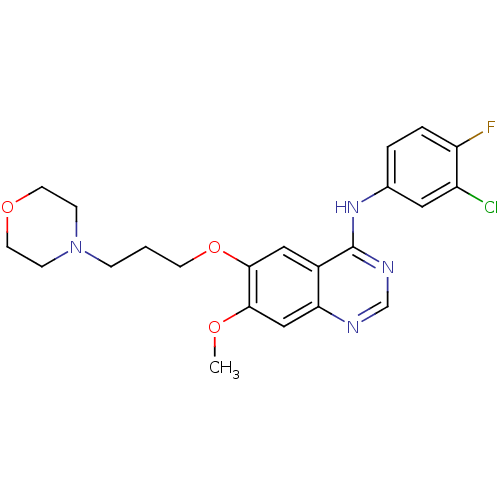
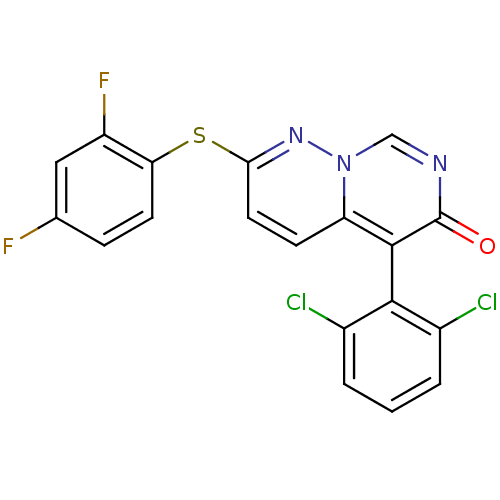
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
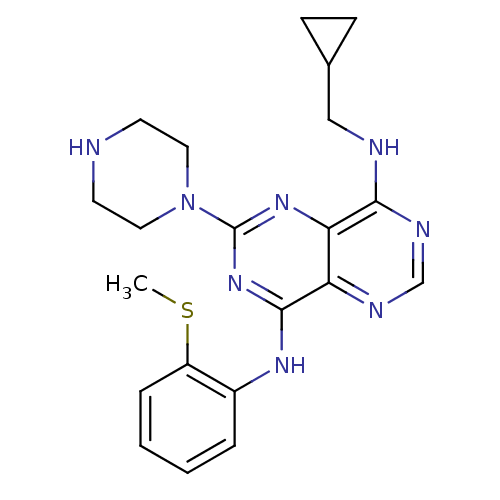
Affinity DataIC50: 1.10nMAssay Description:Inhibition of EphB1 by [gamma33-P]ATP based assayMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
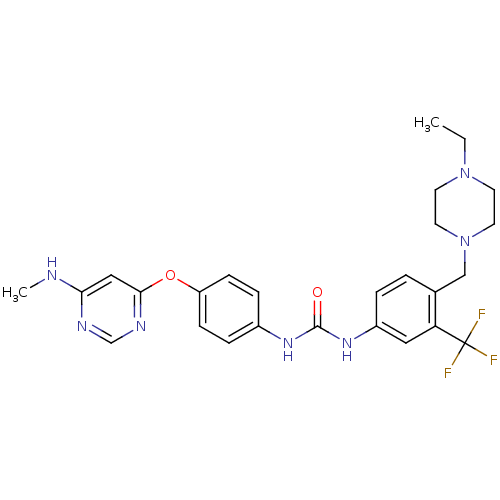
Affinity DataIC50: 20nMAssay Description:Inhibition of human EPHB1 using poly[Glu:Tyr] (4:1) as substrate by [gamma-33P]-ATP assayMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
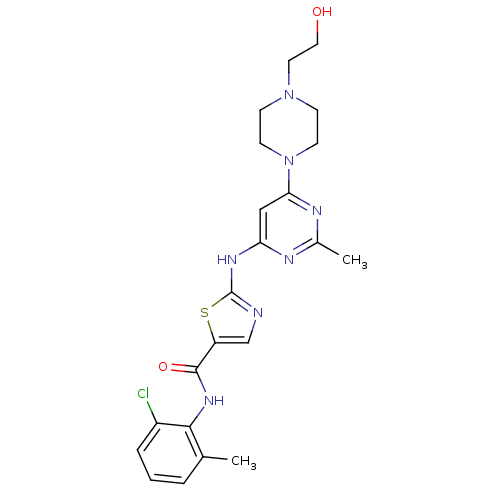
Affinity DataIC50: 30nMAssay Description:Inhibition of EphB1 (unknown origin)More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataIC50: 60.3nMAssay Description:Inhibition of human EPHB1 using poly[Glu:Tyr] (4:1) as substrate by [gamma-33P]-ATP assayMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataIC50: 128nMAssay Description:Inhibition of EPHB1 (unknown origin) incubated for 1 hr by spectrophotometric analysisMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataIC50: 388nMAssay Description:Inhibition of recombinant human EphB1 (564 to end residues) using KVEKIGEGTYGVVYK as substrate incubated for 40 mins in presence of [gamma-33ATP] by ...More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataIC50: 1.11E+3nMAssay Description:Inhibition of EphB1More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataIC50: 3.04E+3nMAssay Description:Inhibition of EPHB1-mediated proliferation of mouse BAF3 cells transformed with TEL-Kinase constructMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataIC50: 3.80E+3nMAssay Description:Displacement of ephrin-B1-Fc from EphB1 receptor Fc ectodomain (unknown origin) after 1 hr by ELISAMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of EphB1 (unknown origin)More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of EPHB1 (unknown origin)More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of human EPHB1 using poly[Glu:Tyr] (4:1) as substrateMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of EPHB1 by FRET assayMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of human EphB1More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataIC50: 1.00E+4nMAssay Description:Inhibition of recombinant human EphB1 (564 to end residues) using KVEKIGEGTYGVV as substrate after 40 mins in presence of [gamma-33ATP] by radiometri...More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataIC50: 3.00E+4nMAssay Description:Inhibition of EPHB1More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataIC50: 3.00E+4nMAssay Description:Inhibition of EPHB1 (unknown origin) incubated for 1 hr by spectrophotometric analysisMore data for this Ligand-Target Pair
Affinity DataIC50: 3.00E+4nMAssay Description:Displacement of biotinylated ephrin-B1-Fc from rat EphB1 Fc preincubated for 1 hr followed by ephrin-B1-FC addition and measured after 4 hrs by ELISAMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataIC50: 3.77E+4nMAssay Description:Inhibition of recombinant human GST-tagged EPHB1 catalytic domain expressed in baculovirus expression system using tyrosine-2 peptide as substrate in...More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataIC50: 4.00E+4nMAssay Description:Inhibition of EPHB1More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 180nMAssay Description:Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity...More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 4.90E+3nMAssay Description:Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity...More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 0.450nMAssay Description:Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity...More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 330nMAssay Description:Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity...More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 3.00E+3nMAssay Description:Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity...More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 240nMAssay Description:Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity...More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 480nMAssay Description:Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity...More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 1.90E+3nMAssay Description:Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity...More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 290nMAssay Description:Kinase inhibitors are a new class of therapeutics with a propensity to inhibit multiple targets. The biological consequences of multi-kinase activity...More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 590nMAssay Description:Average Binding Constant for EPHB1; NA=Not Active at 10 uMMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: >1.00E+4nMAssay Description:Binding affinity to EPHB1More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 5.20E+3nMAssay Description:Binding affinity to EPHB1More data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 240nMAssay Description:Binding constant for EPHB1 kinase domainMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: >1.00E+4nMAssay Description:Binding constant for EPHB1 kinase domainMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 5.20E+3nMAssay Description:Binding constant for EPHB1 kinase domainMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 550nMAssay Description:Binding constant for EPHB1 kinase domainMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 240nMAssay Description:Binding constant for EPHB1 kinase domainMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: >1.00E+4nMAssay Description:Binding constant for EPHB1 kinase domainMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 1.70E+3nMAssay Description:Average Binding Constant for EPHB1; NA=Not Active at 10 uMMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 540nMAssay Description:Average Binding Constant for EPHB1; NA=Not Active at 10 uMMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 1.40E+3nMAssay Description:Average Binding Constant for EPHB1; NA=Not Active at 10 uMMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 960nMAssay Description:Average Binding Constant for EPHB1; NA=Not Active at 10 uMMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 7.30E+3nMAssay Description:Average Binding Constant for EPHB1; NA=Not Active at 10 uMMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 4.20E+3nMAssay Description:Average Binding Constant for EPHB1; NA=Not Active at 10 uMMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: 1.60E+3nMAssay Description:Average Binding Constant for EPHB1; NA=Not Active at 10 uMMore data for this Ligand-Target Pair
TargetEphrin type-B receptor 1(Homo sapiens (Human))
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Genomics Institute of the Novartis Research Foundation
Curated by PDSP Ki Database
Affinity DataKd: >1.00E+4nMAssay Description:Binding affinity to EPHB1More data for this Ligand-Target Pair




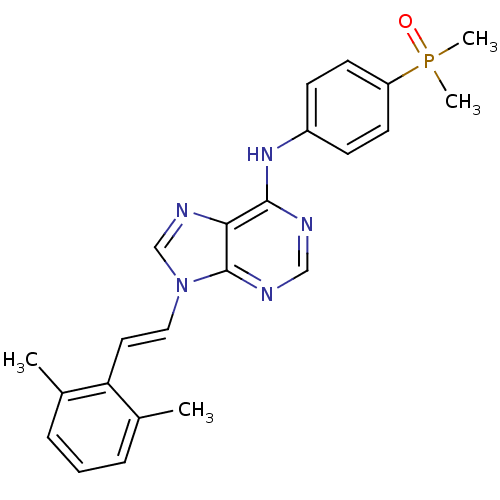

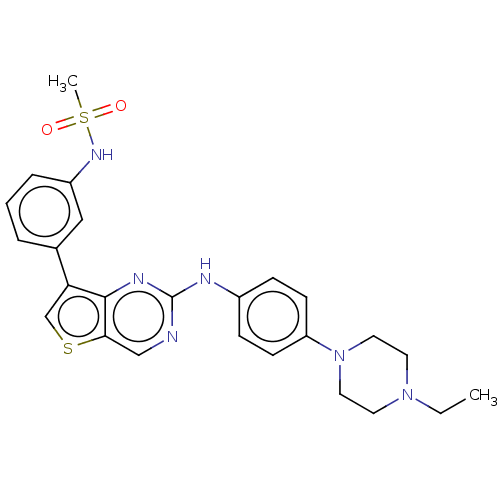

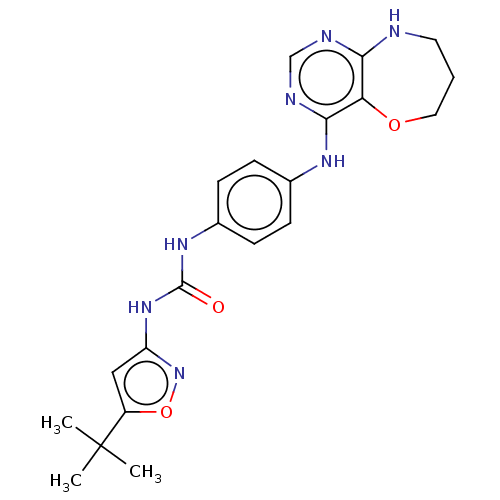
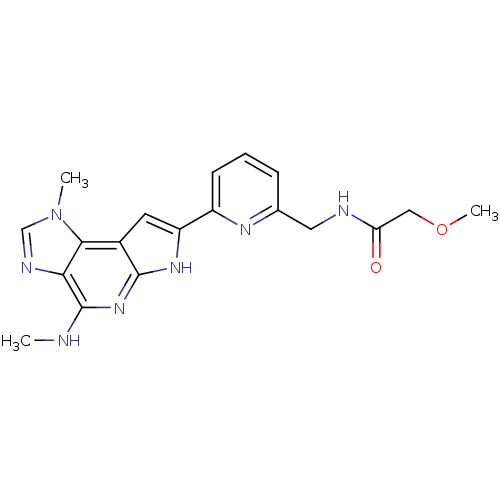
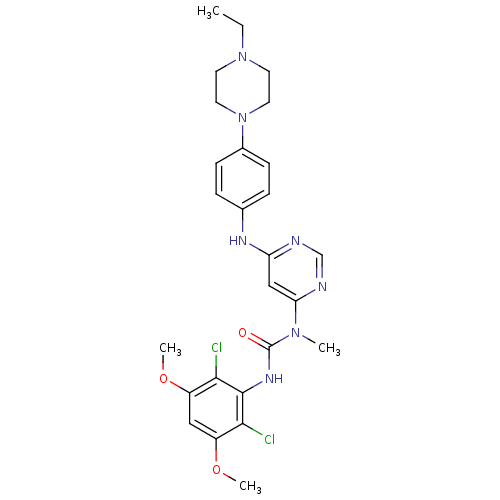
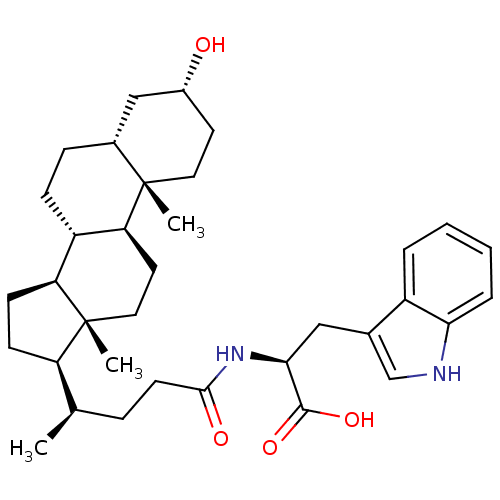
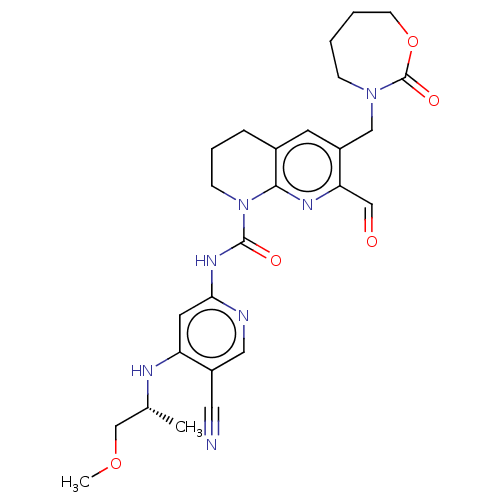
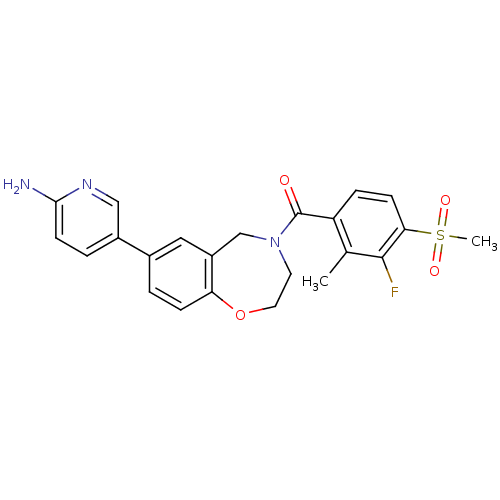
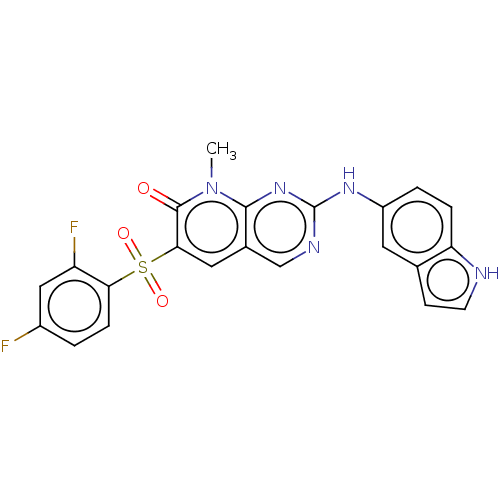
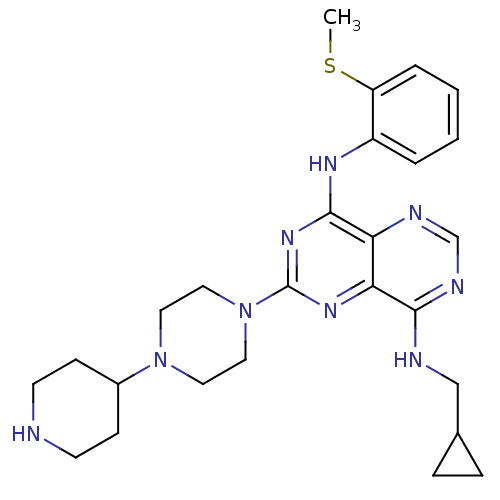

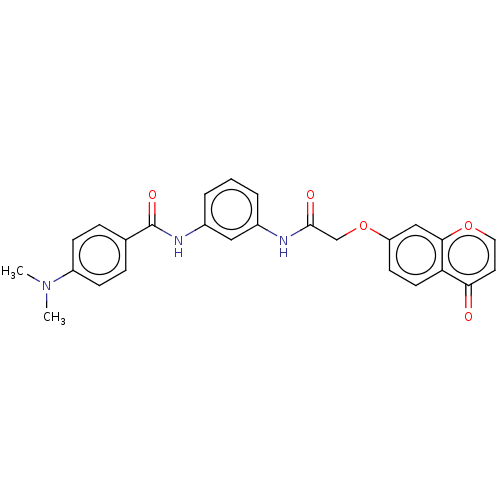

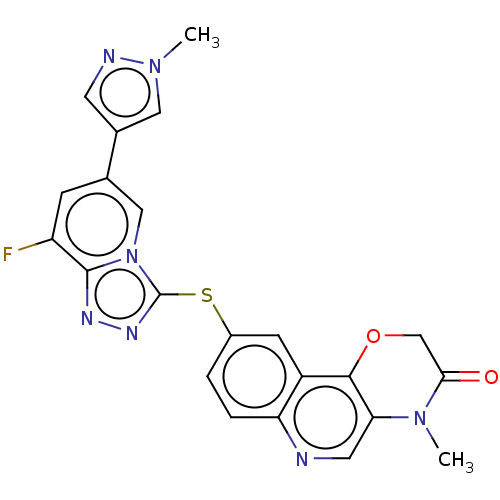
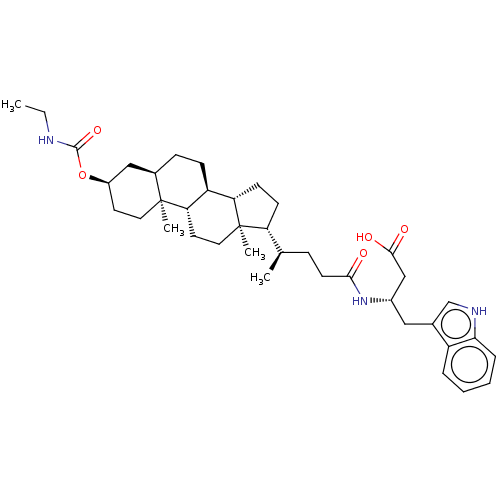
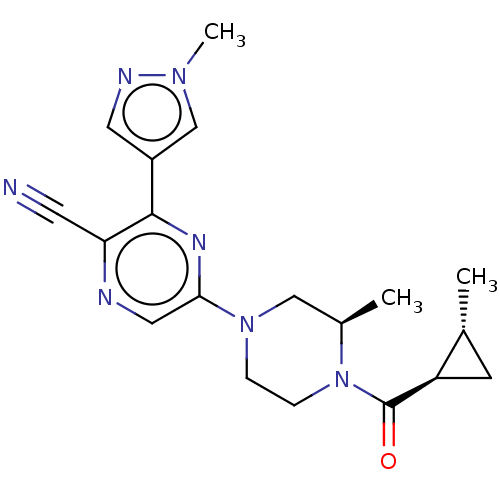
 3D Structure (crystal)
3D Structure (crystal)