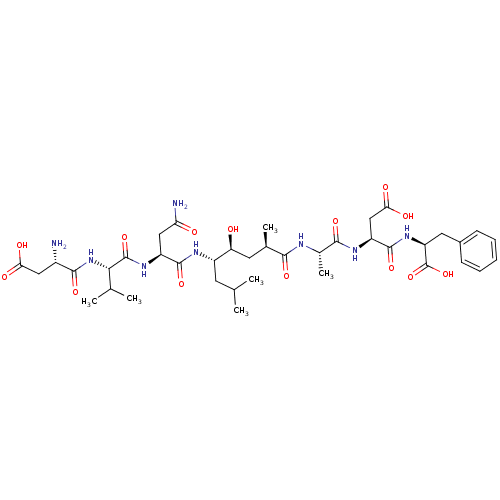
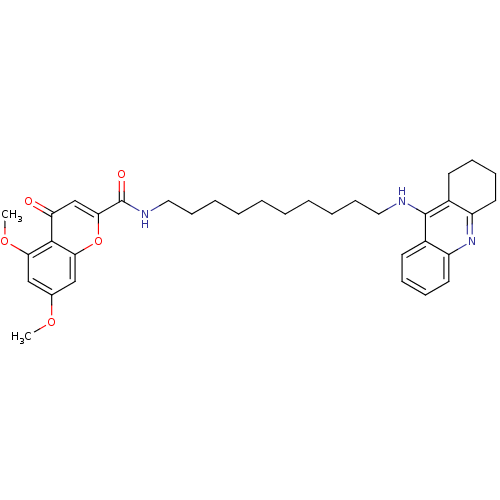
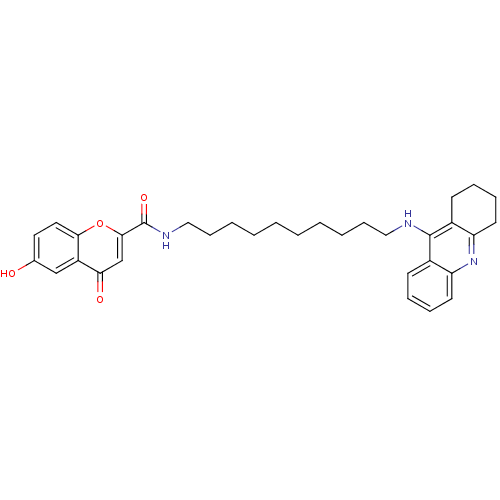
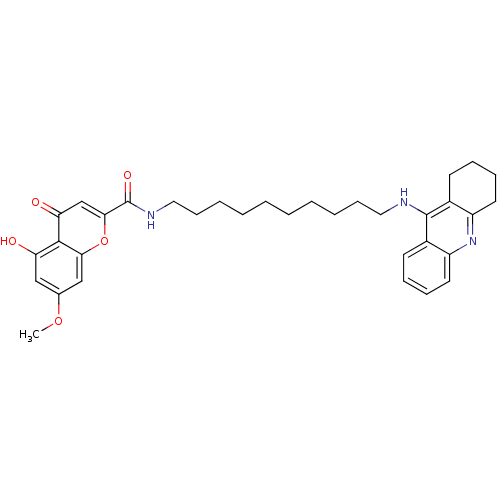
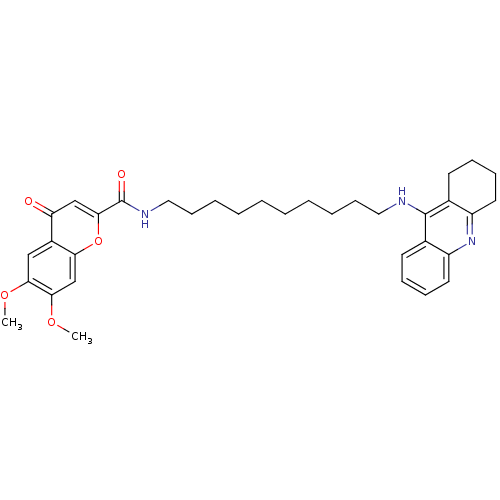
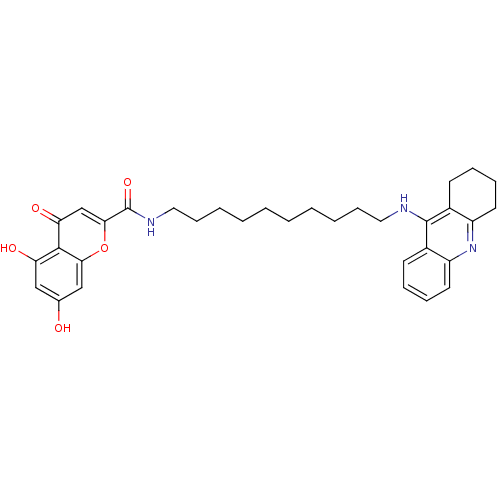
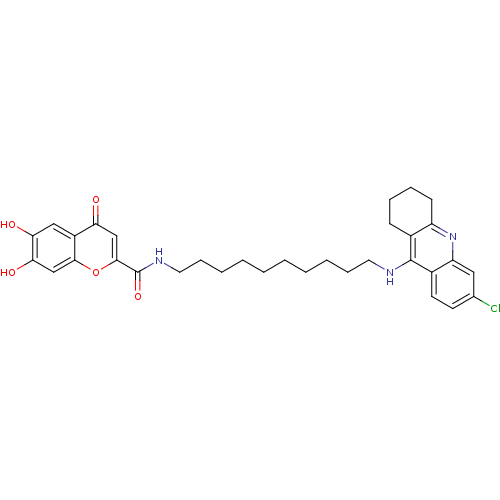
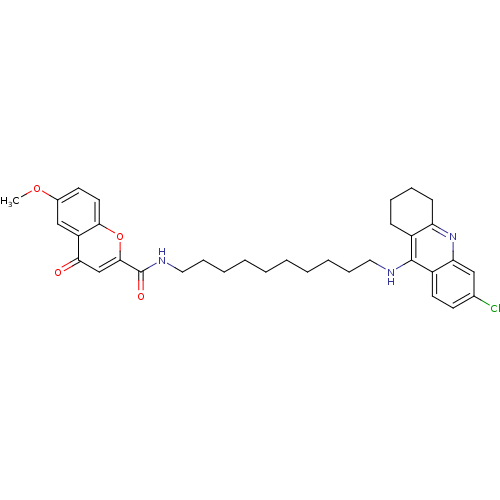
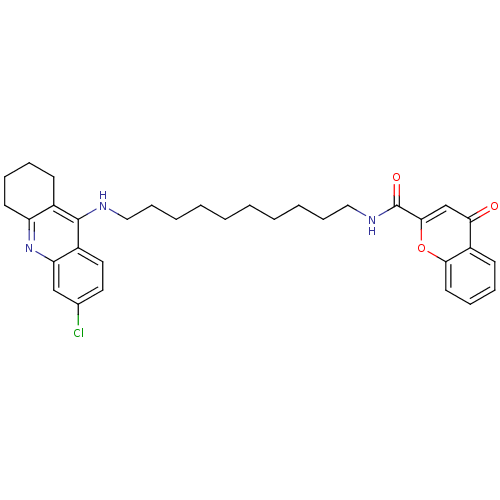
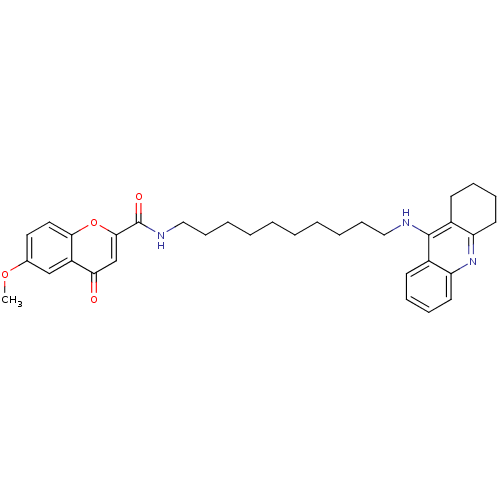
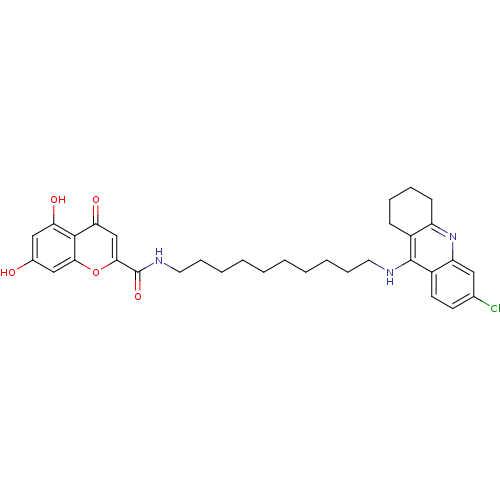
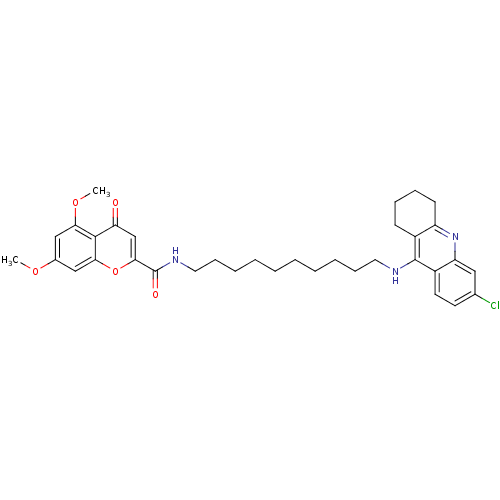
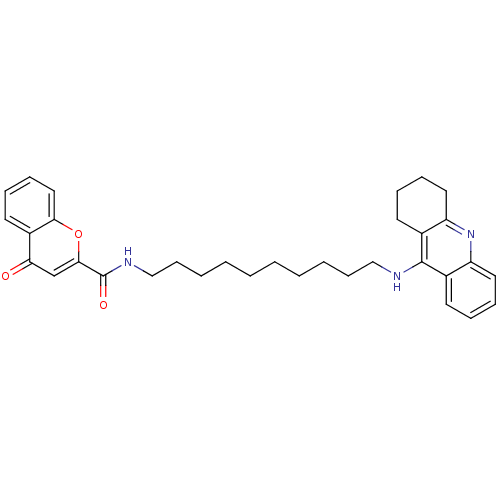
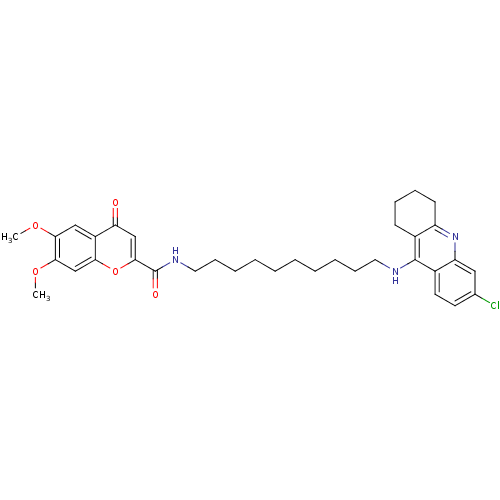
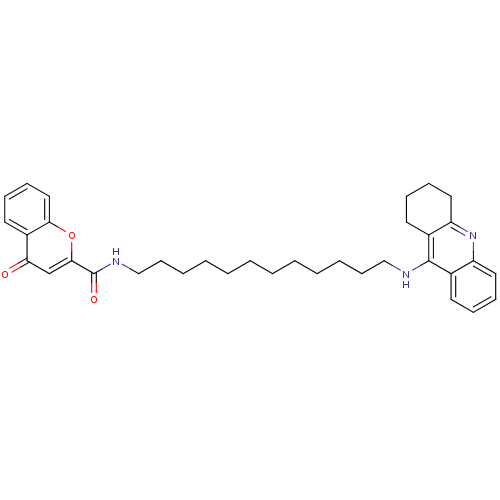
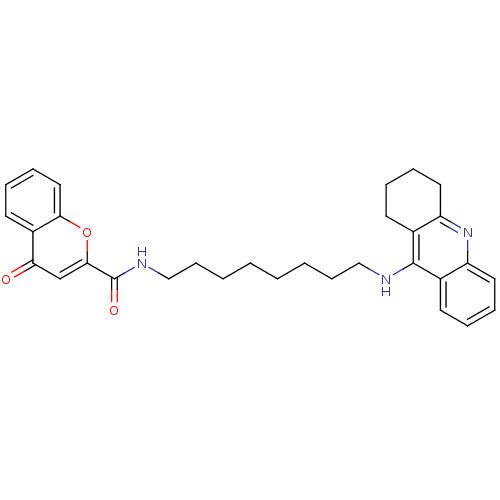
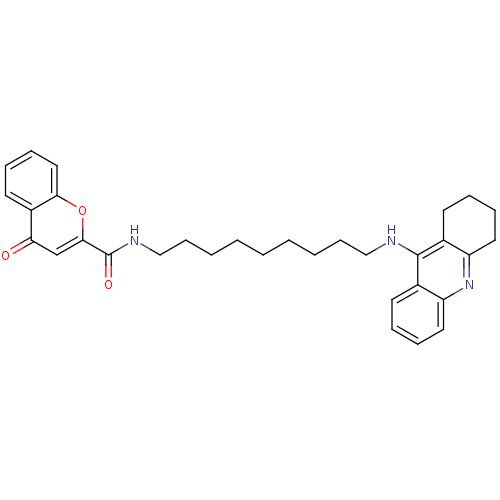
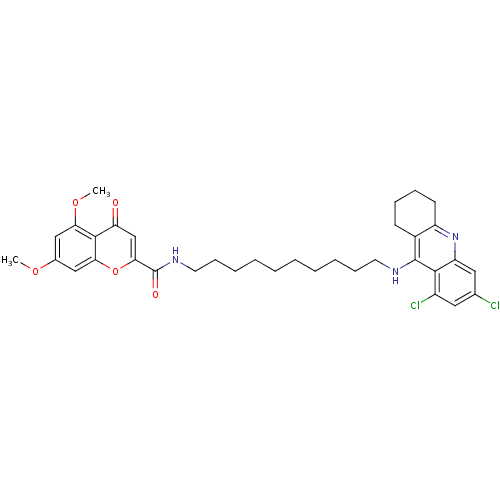
Affinity DataKi: 9.70nMAssay Description:Competitive inhibition of human recombinant BChE using panvera peptide as substrate preincubated for 60 mins before substrate addition by FRET assayMore data for this Ligand-Target Pair
Affinity DataKi: 620nMAssay Description:Competitive inhibition of human recombinant BChE using panvera peptide as substrate preincubated for 60 mins before substrate addition by FRET assayMore data for this Ligand-Target Pair
Affinity DataKi: 820nMAssay Description:Competitive inhibition of human recombinant BChE using panvera peptide as substrate preincubated for 60 mins before substrate addition by FRET assayMore data for this Ligand-Target Pair
Affinity DataKi: 850nMAssay Description:Competitive inhibition of human recombinant BChE using panvera peptide as substrate preincubated for 60 mins before substrate addition by FRET assayMore data for this Ligand-Target Pair
Affinity DataKi: 880nMAssay Description:Competitive inhibition of human recombinant BChE using panvera peptide as substrate preincubated for 60 mins before substrate addition by FRET assayMore data for this Ligand-Target Pair
Affinity DataKi: 1.06E+3nMAssay Description:Competitive inhibition of human recombinant BChE using panvera peptide as substrate preincubated for 60 mins before substrate addition by FRET assayMore data for this Ligand-Target Pair
Affinity DataKi: 3.99E+3nMAssay Description:Competitive inhibition of human recombinant BChE using panvera peptide as substrate preincubated for 60 mins before substrate addition by FRET assayMore data for this Ligand-Target Pair
Affinity DataKi: 4.23E+3nMAssay Description:Competitive inhibition of human recombinant BChE using panvera peptide as substrate preincubated for 60 mins before substrate addition by FRET assayMore data for this Ligand-Target Pair
Affinity DataKi: 6.58E+3nMAssay Description:Competitive inhibition of human recombinant BChE using panvera peptide as substrate preincubated for 60 mins before substrate addition by FRET assayMore data for this Ligand-Target Pair
Affinity DataKi: 1.13E+4nMAssay Description:Competitive inhibition of human recombinant BChE using panvera peptide as substrate preincubated for 60 mins before substrate addition by FRET assayMore data for this Ligand-Target Pair
Affinity DataIC50: 0.0350nMAssay Description:Inhibition of human erythrocytes AChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.0400nMAssay Description:Inhibition of human serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.0700nMAssay Description:Inhibition of human erythrocytes AChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.0800nMAssay Description:Inhibition of human serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.0900nMAssay Description:Inhibition of human erythrocytes AChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.100nMAssay Description:Inhibition of human serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.100nMAssay Description:Inhibition of human serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.100nMAssay Description:Inhibition of human erythrocytes AChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.100nMAssay Description:Inhibition of human serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.150nMAssay Description:Inhibition of human serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.180nMAssay Description:Inhibition of horse serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.180nMAssay Description:Inhibition of horse serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.180nMAssay Description:Inhibition of horse serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.200nMAssay Description:Inhibition of human erythrocytes AChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.25nMAssay Description:Inhibition of horse serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.300nMAssay Description:Inhibition of human erythrocytes AChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.320nMAssay Description:Inhibition of horse serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.330nMAssay Description:Inhibition of horse serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.350nMAssay Description:Inhibition of horse serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.350nMAssay Description:Inhibition of human erythrocytes AChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.430nMAssay Description:Inhibition of horse serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.5nMAssay Description:Inhibition of horse serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.550nMAssay Description:Inhibition of horse serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.580nMAssay Description:Inhibition of horse serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.75nMAssay Description:Inhibition of horse serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.780nMAssay Description:Inhibition of human erythrocytes AChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.800nMAssay Description:Inhibition of human serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 0.850nMAssay Description:Inhibition of horse serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of human erythrocytes AChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of human serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of horse serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of horse serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1nMAssay Description:Inhibition of human erythrocytes AChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMAssay Description:Inhibition of human serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1.5nMAssay Description:Inhibition of human serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1.80nMAssay Description:Inhibition of human erythrocytes AChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 1.80nMAssay Description:Inhibition of horse serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 2nMAssay Description:Inhibition of horse serum BChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 2.30nMAssay Description:Inhibition of human erythrocytes AChE by Ellman's methodMore data for this Ligand-Target Pair
Affinity DataIC50: 2.30nMAssay Description:Inhibition of human erythrocytes AChE by Ellman's methodMore data for this Ligand-Target Pair