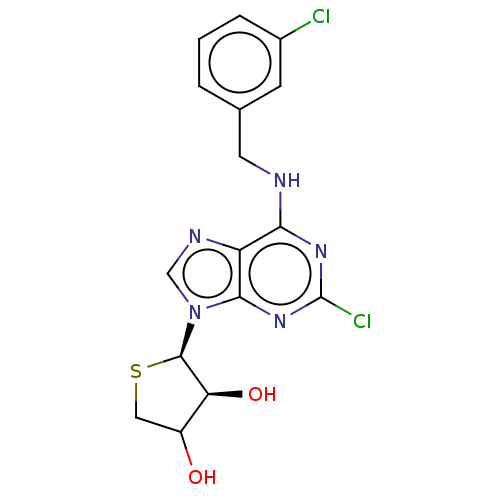
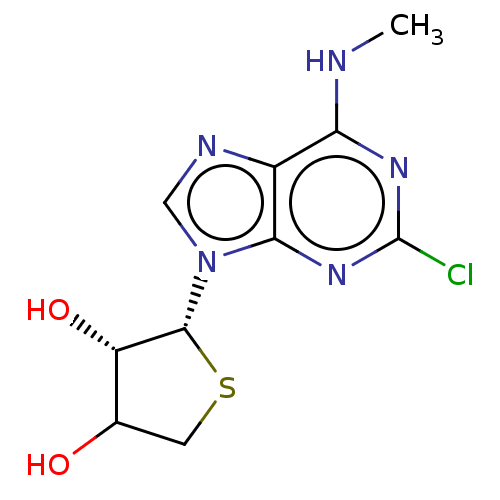
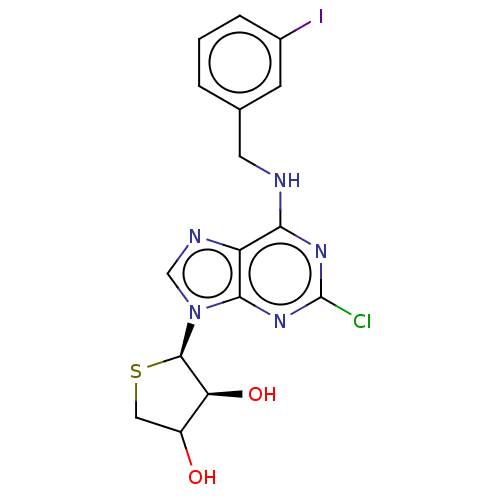
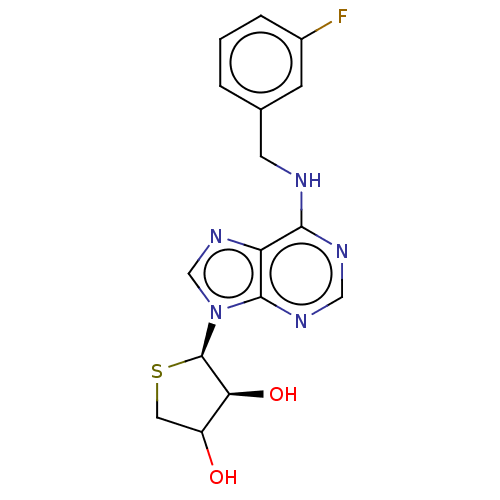
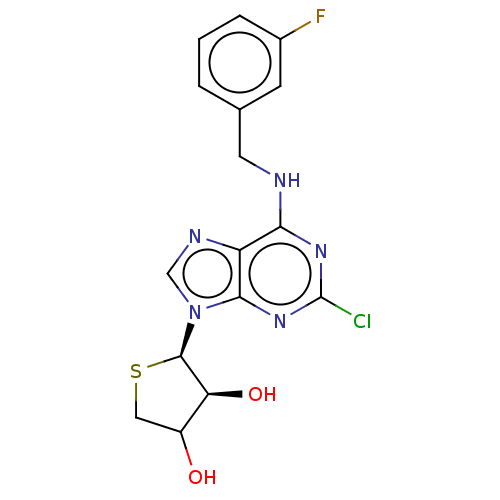
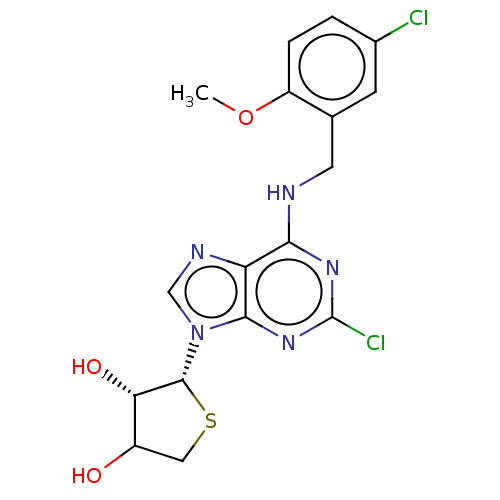
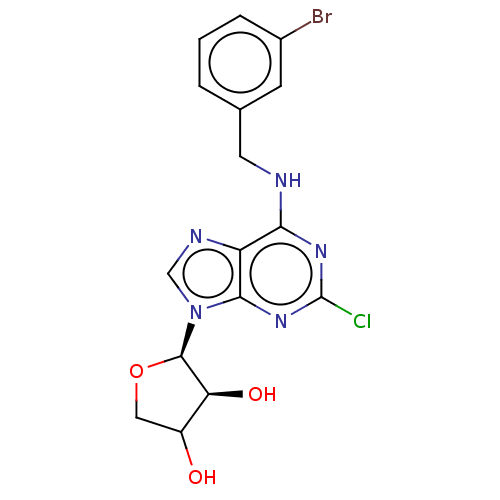
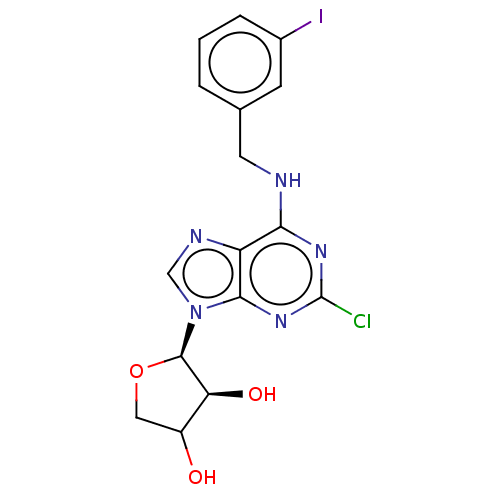
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 1.5nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 1.66nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 2.5nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 3.69nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 4.16nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 6.80nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 7.30nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 7.40nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 8.99nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 12.7nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 13nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 19.9nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 24.8nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 25.8nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 42.9nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 230nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 341nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 420nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 440nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A1(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 530nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A1(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 790nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A1(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 860nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 1.26E+3nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A1(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 1.43E+3nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 1.60E+3nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A1(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 2.49E+3nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair
TargetAdenosine receptor A2a(Homo sapiens (Human))
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
FM Therapeutics Co., Ltd.; The United States of America, As Represented by the Secretary, Department of Health and Human Services, The Office of Technology Transfer, National Institutes of Health
US Patent
Affinity DataKi: 4.02E+3nMAssay Description:CHO cells (ATCC No. CCL-61), in which A1 and A3 adenosine receptors were expressed, were cultured in F-12 media (Gibco, U.S.A.) supplemented with 10%...More data for this Ligand-Target Pair