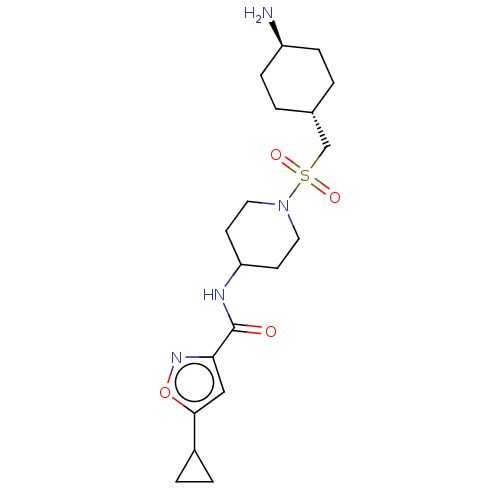
Reaction Details  Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Histone-lysine N-methyltransferase SMYD3
Ligand
BDBM50509572
Substrate
n/a
Meas. Tech.
ChEMBL_1838817 (CHEMBL4338950)
IC50
2.5±n/a nM
Citation
 Su, DS; Qu, J; Schulz, M; Blackledge, CW; Yu, H; Zeng, J; Burgess, J; Reif, A; Stern, M; Nagarajan, R; Pappalardi, MB; Wong, K; Graves, AP; Bonnette, W; Wang, L; Elkins, P; Knapp-Reed, B; Carson, JD; McHugh, C; Mohammad, H; Kruger, R; Luengo, J; Heerding, DA; Creasy, CL Discovery of Isoxazole Amides as Potent and Selective SMYD3 Inhibitors. ACS Med Chem Lett 11:133-140 (2020) [PubMed] Article
Su, DS; Qu, J; Schulz, M; Blackledge, CW; Yu, H; Zeng, J; Burgess, J; Reif, A; Stern, M; Nagarajan, R; Pappalardi, MB; Wong, K; Graves, AP; Bonnette, W; Wang, L; Elkins, P; Knapp-Reed, B; Carson, JD; McHugh, C; Mohammad, H; Kruger, R; Luengo, J; Heerding, DA; Creasy, CL Discovery of Isoxazole Amides as Potent and Selective SMYD3 Inhibitors. ACS Med Chem Lett 11:133-140 (2020) [PubMed] ArticleMore Info.:
Target
Name:
Histone-lysine N-methyltransferase SMYD3
Synonyms:
SET and MYND domain-containing protein 3 | SMYD3 | SMYD3_HUMAN | ZMYND1 | ZNFN3A1 | Zinc finger MYND domain-containing protein 1
Type:
Enzyme
Mol. Mass.:
49101.22
Organism:
Homo sapiens (Human)
Description:
Q9H7B4-2
Residue:
428
Sequence:
MEPLKVEKFATAKRGNGLRAVTPLRPGELLFRSDPLAYTVCKGSRGVVCDRCLLGKEKLMRCSQCRVAKYCSAKCQKKAWPDHKRECKCLKSCKPRYPPDSVRLLGRVVFKLMDGAPSESEKLYSFYDLESNINKLTEDKKEGLRQLVMTFQHFMREEIQDASQLPPAFDLFEAFAKVICNSFTICNAEMQEVGVGLYPSISLLNHSCDPNCSIVFNGPHLLLRAVRDIEVGEELTICYLDMLMTSEERRKQLRDQYCFECDCFRCQTQDKDADMLTGDEQVWKEVQESLKKIEELKAHWKWEQVLAMCQAIISSNSERLPDINIYQLKVLDCAMDACINLGLLEEALFYGTRTMEPYRIFFPGSHPVRGVQVMKVGKLQLHQGMFPQAMKNLRLAFDIMRVTHGREHSLIEDLILLLEECDANIRAS
Inhibitor
Name:
BDBM50509572
Synonyms:
CHEMBL4521928
Type:
Small organic molecule
Emp. Form.:
C19H30N4O4S
Mol. Mass.:
410.531
SMILES:
N[C@H]1CC[C@H](CS(=O)(=O)N2CCC(CC2)NC(=O)c2cc(on2)C2CC2)CC1 |r,wU:4.4,wD:1.0,(21.16,-1.33,;19.82,-2.09,;18.49,-1.31,;17.14,-2.07,;17.14,-3.6,;15.8,-4.37,;14.47,-3.59,;13.69,-2.25,;15.24,-2.25,;13.13,-4.37,;11.8,-3.58,;10.48,-4.37,;10.48,-5.91,;11.8,-6.66,;13.13,-5.91,;9.15,-6.67,;7.81,-5.91,;7.81,-4.37,;6.48,-6.68,;6.33,-8.21,;4.82,-8.53,;4.04,-7.21,;5.07,-6.06,;4.2,-9.94,;2.95,-10.85,;4.36,-11.47,;18.47,-4.39,;19.81,-3.63,)|