Reaction Details  Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Orexin/Hypocretin receptor type 1
Ligand
BDBM386067
Substrate
n/a
Meas. Tech.
ChEMBL_2165224 (CHEMBL5050085)
EC50
>100000±n/a nM
Citation
 Fujimoto, T; Rikimaru, K; Fukuda, K; Sugimoto, H; Masuda, K; Ohyabu, N; Banno, Y; Tokunaga, N; Kawamoto, T; Tomata, Y; Kumagai, Y; Iida, M; Nagano, Y; Yoneyama-Hirozane, M; Shimizu, Y; Sasa, K; Ishikawa, T; Yukitake, H; Ito, M; Aoyama, K; Matsumoto, T Discovery of TAK-925 as a Potent, Selective, and Brain-Penetrant Orexin 2 Receptor Agonist. ACS Med Chem Lett 13:457-462 (2022) [PubMed] Article
Fujimoto, T; Rikimaru, K; Fukuda, K; Sugimoto, H; Masuda, K; Ohyabu, N; Banno, Y; Tokunaga, N; Kawamoto, T; Tomata, Y; Kumagai, Y; Iida, M; Nagano, Y; Yoneyama-Hirozane, M; Shimizu, Y; Sasa, K; Ishikawa, T; Yukitake, H; Ito, M; Aoyama, K; Matsumoto, T Discovery of TAK-925 as a Potent, Selective, and Brain-Penetrant Orexin 2 Receptor Agonist. ACS Med Chem Lett 13:457-462 (2022) [PubMed] ArticleMore Info.:
Target
Name:
Orexin/Hypocretin receptor type 1
Synonyms:
HCRTR1 | Hypocretin receptor type 1 | OX1R_HUMAN | Orexin receptor type 1 | Orexin receptor type 1 (OR 1) | Orexin receptor type 1 (OR-1) | Orexin receptor type 1 (OX1) | Orexin receptor type 1 (OX1R) | Orexin receptor type 1 (OxR1) | Ox1r
Type:
Protein
Mol. Mass.:
47554.50
Organism:
Homo sapiens (Human)
Description:
O43613
Residue:
425
Sequence:
MEPSATPGAQMGVPPGSREPSPVPPDYEDEFLRYLWRDYLYPKQYEWVLIAAYVAVFVVALVGNTLVCLAVWRNHHMRTVTNYFIVNLSLADVLVTAICLPASLLVDITESWLFGHALCKVIPYLQAVSVSVAVLTLSFIALDRWYAICHPLLFKSTARRARGSILGIWAVSLAIMVPQAAVMECSSVLPELANRTRLFSVCDERWADDLYPKIYHSCFFIVTYLAPLGLMAMAYFQIFRKLWGRQIPGTTSALVRNWKRPSDQLGDLEQGLSGEPQPRARAFLAEVKQMRARRKTAKMLMVVLLVFALCYLPISVLNVLKRVFGMFRQASDREAVYACFTFSHWLVYANSAANPIIYNFLSGKFREQFKAAFSCCLPGLGPCGSLKAPSPRSSASHKSLSLQSRCSISKISEHVVLTSVTTVLP
Inhibitor
Name:
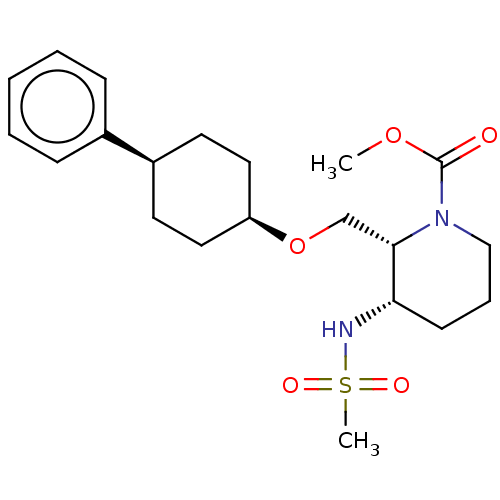
BDBM386067
Synonyms:
US10287305, Example 5 | US10508083, Example 191 | US11292766, Example 191 | methyl (2R,3S)-3- ((methylsulfonyl)amino)-2-(((cis-4- phenylcyclohexyl)oxy)methyl)- piperidine-1-carboxylate
Type:
Small organic molecule
Emp. Form.:
C21H32N2O5S
Mol. Mass.:
424.554
SMILES:
COC(=O)N1CCC[C@H](NS(C)(=O)=O)[C@@H]1CO[C@H]1CC[C@H](CC1)c1ccccc1 |r,wU:20.24,17.17,14.15,8.8,(-1.33,-4.23,;,-3.47,;1.33,-4.23,;1.33,-5.78,;2.67,-3.47,;4,-4.24,;5.33,-3.47,;5.33,-1.93,;4,-1.16,;4,.38,;5.33,1.15,;6.67,.38,;4.56,2.49,;6.1,2.49,;2.67,-1.93,;1.33,-1.16,;1.33,.38,;,1.15,;-1.33,.38,;-2.67,1.15,;-2.67,2.69,;-1.33,3.46,;,2.69,;-4,3.46,;-4,5,;-5.33,5.78,;-6.67,5,;-6.67,3.46,;-5.33,2.69,)|