Reaction Details  Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Bifunctional purine biosynthesis protein ATIC
Ligand
BDBM50167703
Substrate
n/a
Meas. Tech.
ChEMBL_302287 (CHEMBL830304)
Ki
3100±n/a nM
Citation
 Capps, KJ; Humiston, J; Dominique, R; Hwang, I; Boger, DL Discovery of AICAR Tfase inhibitors that disrupt requisite enzyme dimerization. Bioorg Med Chem Lett 15:2840-4 (2005) [PubMed] Article
Capps, KJ; Humiston, J; Dominique, R; Hwang, I; Boger, DL Discovery of AICAR Tfase inhibitors that disrupt requisite enzyme dimerization. Bioorg Med Chem Lett 15:2840-4 (2005) [PubMed] ArticleMore Info.:
Target
Name:
Bifunctional purine biosynthesis protein ATIC
Synonyms:
5-aminoimidazole-4-carboxamide ribonucleotide formyltransferase | 5-aminoimidazole-4-carboxamide-ribonucleotide transformylase | AICAR Tfase | AICAR transformylase | ATIC | Aminoimidazole carboxamide ribonucleotide transformylase (AICAR Tfase) | Bifunctional purine biosynthesis protein PURH | IMP Cyclohydrolase (IMPCH) | IMP cyclohydrolase | IMP synthetase | Inosinicase | PUR9_HUMAN | PURH | Phosphoribosylaminoimidazolecarboxamide formyltransferase | Thymidylate synthase/GAR transformylase/AICAR transformylase
Type:
Protein
Mol. Mass.:
64616.62
Organism:
Homo sapiens (Human)
Description:
P31939
Residue:
592
Sequence:
MAPGQLALFSVSDKTGLVEFARNLTALGLNLVASGGTAKALRDAGLAVRDVSELTGFPEMLGGRVKTLHPAVHAGILARNIPEDNADMARLDFNLIRVVACNLYPFVKTVASPGVTVEEAVEQIDIGGVTLLRAAAKNHARVTVVCEPEDYVVVSTEMQSSESKDTSLETRRQLALKAFTHTAQYDEAISDYFRKQYSKGVSQMPLRYGMNPHQTPAQLYTLQPKLPITVLNGAPGFINLCDALNAWQLVKELKEALGIPAAASFKHVSPAGAAVGIPLSEDEAKVCMVYDLYKTLTPISAAYARARGADRMSSFGDFVALSDVCDVPTAKIISREVSDGIIAPGYEEEALTILSKKKNGNYCVLQMDQSYKPDENEVRTLFGLHLSQKRNNGVVDKSLFSNVVTKNKDLPESALRDLIVATIAVKYTQSNSVCYAKNGQVIGIGAGQQSRIHCTRLAGDKANYWWLRHHPQVLSMKFKTGVKRAEISNAIDQYVTGTIGEDEDLIKWKALFEEVPELLTEAEKKEWVEKLTEVSISSDAFFPFRDNVDRAKRSGVAYIAAPSGSAADKVVIEACDELGIILAHTNLRLFHH
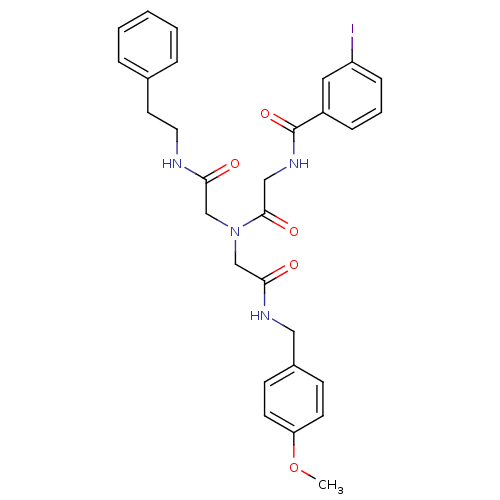
Inhibitor
Name:
BDBM50167703
Synonyms:
3-Iodo-N-{[[(4-methoxy-benzylcarbamoyl)-methyl]-(phenethylcarbamoyl-methyl)-carbamoyl]-methyl}-benzamide | CHEMBL193445
Type:
Small organic molecule
Emp. Form.:
C29H31IN4O5
Mol. Mass.:
642.4847
SMILES:
COc1ccc(CNC(=O)CN(CC(=O)NCCc2ccccc2)C(=O)CNC(=O)c2cccc(I)c2)cc1