Reaction Details  Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Carboxylic ester hydrolase
Ligand
BDBM10750
Substrate
n/a
Meas. Tech.
ChEMBL_877980 (CHEMBL2189046)
IC50
480±n/a nM
Citation
 Yanovsky, I; Finkin-Groner, E; Zaikin, A; Lerman, L; Shalom, H; Zeeli, S; Weill, T; Ginsburg, I; Nudelman, A; Weinstock, M Carbamate derivatives of indolines as cholinesterase inhibitors and antioxidants for the treatment of Alzheimer's disease. J Med Chem 55:10700-15 (2012) [PubMed] Article
Yanovsky, I; Finkin-Groner, E; Zaikin, A; Lerman, L; Shalom, H; Zeeli, S; Weill, T; Ginsburg, I; Nudelman, A; Weinstock, M Carbamate derivatives of indolines as cholinesterase inhibitors and antioxidants for the treatment of Alzheimer's disease. J Med Chem 55:10700-15 (2012) [PubMed] ArticleMore Info.:
Target
Name:
Carboxylic ester hydrolase
Synonyms:
BuChE | Butyrlcholinesterase (BuChE) | Butyrylcholine esterase | Butyrylcholinesterase | Butyrylcholinesterase (BChE) | Butyrylcholinesterase (BuChE) | Butyrylcholinesterase (EqBuChE) | Carboxylic ester hydrolase | butyrylcholinesterase precursor
Type:
Protein
Mol. Mass.:
68842.83
Organism:
Equus caballus (Horse)
Description:
Q9N1N9
Residue:
602
Sequence:
MQSWGTIICIRILLRFLLLWVLIGNSHTEEDIIITTKNGKVRGMNLPVLGGTVTAFLGIPYAQPPLGRLRFKKPQSLTKWSNIWNATKYANSCYQNTDQSFPGFLGSEMWNPNTELSEDCLYLNVWIPAPKPKNATVMIWIYGGGFQTGTSSLPVYDGKFLARVERVIVVSMNYRVGALGFLALSENPEAPGNMGLFDQQLALQWVQKNIAAFGGNPRSVTLFGESAGAASVSLHLLSPRSQPLFTRAILQSGSSNAPWAVTSLYEARNRTLTLAKRMGCSRDNETEMIKCLRDKDPQEILLNEVFVVPYDTLLSVNFGPTVDGDFLTDMPDTLLQLGQFKRTQILVGVNKDEGTAFLVYGAPGFSKDNNSIITRKEFQEGLKIFFPRVSEFGRESILFHYMDWLDDQRAENYREALDDVVGDYNIICPALEFTKKFSELGNDAFFYYFEHRSTKLPWPEWMGVMHGYEIEFVFGLPLERRVNYTKAEEILSRSIMKRWANFAKYGNPNGTQSNSTRWPVFKSTEQKYLTLNTESPKVYTKLRAQQCRFWTLFFPKVLELTGNIDEAEREWKAGFHRWNNYMMDWKNQFNDYTSKKESCSDF
Inhibitor
Name:
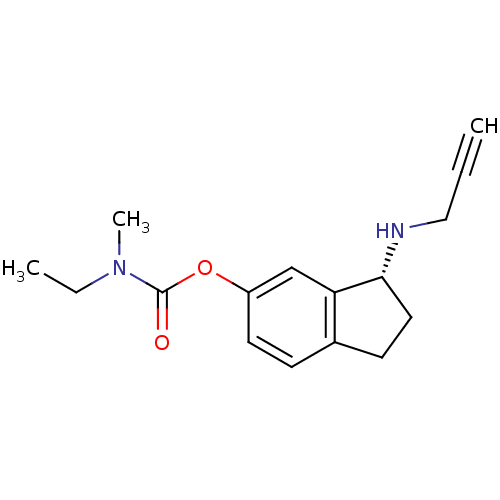
BDBM10750
Synonyms:
(3R)-3-(prop-2-yn-1-ylamino)-2,3-dihydro-1H-inden-5-yl N-ethyl-N-methylcarbamate hydrochloride | Aminoindan deriv. (R)18b | Aminoindan deriv. (R)18b.HCl | N-propargylaminoindan (R)18b.HCl | R-CPAI
Type:
Small organic molecule
Emp. Form.:
C16H20N2O2
Mol. Mass.:
272.3422
SMILES:
CCN(C)C(=O)Oc1ccc2CC[C@@H](NCC#C)c2c1 |r|