Reaction Details  Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Adhesin Ata autotransporter
Ligand
BDBM281023
Substrate
n/a
Meas. Tech.
Antimicrobial Susceptibility Testing Assay
IC50
1000±n/a nM
Citation
 Alanine, A; Beignet, J; Bleicher, K; Fasching, B; Hilpert, H; Hu, T; MacDonald, D; Jackson, S; Kolczewski, S; Kroll, C; Schaeublin, A; Shen, H; Stoll, T; Thomas, H; Wahhab, A; Zampaloni, C Peptide macrocycles against acinetobacter baumannii US Patent US10030047 Publication Date 7/24/2018
Alanine, A; Beignet, J; Bleicher, K; Fasching, B; Hilpert, H; Hu, T; MacDonald, D; Jackson, S; Kolczewski, S; Kroll, C; Schaeublin, A; Shen, H; Stoll, T; Thomas, H; Wahhab, A; Zampaloni, C Peptide macrocycles against acinetobacter baumannii US Patent US10030047 Publication Date 7/24/2018More Info.:
Target
Name:
Adhesin Ata autotransporter
Synonyms:
ATA_ACIBT | Acinetobacter trimeric autotransporter | ata | ata:
Type:
Enzyme Catalytic Domain
Mol. Mass.:
189662.81
Organism:
Acinetobacter baumannii (strain ATCC 17978 / CIP 53.77 / LMG 1025 / NCDC KC755 / 5377)
Description:
A3M3H0
Residue:
1873
Sequence:
MNKVYKVIWNASIGAWVATSEIAKSKTKTKSKTLNLSAAVLSGVICFAPNAFAGTNTEGGIGQGTSISGTTSCREGSANTANQKDIAIGCGAQTQDRTGSNIANRNNPYNNSTGAYAGAMKQGGAISVGTGAVVEKGLGTAIGSYATTQGISGVAIGTGALSSGNTALAVGRQSAATADFSQAIGNVAAATGKGSLAIGHSATAEGYRSIAIGSPDIENADPVAGQAGAAYQPKMATKATGKDSIAFGGGAVATEENALAIGAFSESKGKKSVAIGTGAKAQKDNAVVIGDQAEASFEGGVAIGKGARSEAENSIALGKDSKASQATGESFLTKQSAPTGVLSIGDIGTERRIQNVADGAADSDAATVRQLKAARTHYVSINDNGQPGGNFENDGATGRNAIAVGVNASAAGREAMAIGGNAQAIGSGAIAMGSSSQTVGRGDVAIGRNASTQGAEGVNSNQSVAIGDQTKAIGDQSVAIGADVIAKGNSSVAIGGDDVDKIARDTELSNTYTEITGGTLQAGKYPTTEANHGSTAVGVQAVGTGAFSSAFGMTSKATGDASSAFGVMSNASGKGAAAFGAVAQATGDGASAMGINSLASGTNSTAIGSGNKPGEGANATGNSSAAIGSGAQATGDNSAAIGKGAEATNENAAAVGGGAKATGKNAAAIGGGAIADQENAVAVGQGAQSLVEGGVALGARSKVEAKNSVALGQDAVATEATGTSFLTNRDASQSNGVISVGSAGKERRITNVEDGSADSDAVTVRQLKNVDSRVNQNTSNIGKNTQNITNLNQKLDDTKTNLGNQIADTNKNLNDAKKDLGNQITDTNTKLNTTKDQLTTQINDTNTELNNTIGNTKTELNTKIDNTKTELENKGLNFAGNSGADVHRKLGDKLNIVGGAAASTPAAKTSGENVITRTTQDGIQIELLKDSKFDSVTTGNTTLNTNGLTIKEGPSITKQGINAGSKQISNVADGINAKDAVNVDQLTKVKDNLNGRITDTNNQLNDAKKDLGNQIADTNKNLNDAKKDLGNQITDTNTKLNNTKDQLTTQINDTKTELNNTIGNTKTELNSKIDSTKTELENKGLNFAGNSGADVHRKLGEKLNIIGGAAASTPAAKTSGENVITRTTKDGIQIELLKDSKFDSVTTGNTTLNTNGLTIKEGPSITKDGINASGKQITNVADGVNAKDAVNKGQLDNLAAKQNATDDAAVKYDDAKTKDKVTLKGKDGTVLDNVKAGHISSTSKEAVNGSQIHNISNSIKNSIGGNTVVNPDGSLTTNNIGGTGKNNINDAISEVKNTATKAKTTVTEGDNIVVKETVNKDGSTNYEVSTKKDLTLNSVTTGDTVLNNNGLTIKDGPSITKDGVNAGGKKITDVANGVIAQNSKDAVNGAQVHHISNSIKNSIGGNTVVNPDGSLTTNNIGGTGKNNINDAIKSVDEKVTNGVNDLTQKGLNFGANDQKTTQGKAVHRKLGDTINIVGGADAKTAEDKTSGENIITRTTEDGVKIEMLKDVKFDSVNVGGHVLNQQGLIIKGGPSITVNGINAGGKQITNVADGINAKDAVNKGQLDKQINEVKDQIGKDIGKLSDHAVQYDKDKNGNVDKSSVTLGGGEKGTNLKNVADGKVAEGSKDAVNGGQLWNVQNQVDKNSNDIKNIQNNIDNISNGKAGLVQQQKPNGEITVGRDTGGTSINMAGKEGDRVVQGVKDGEIKAGSNQAVNGGQIHKISESIKNSIGGNTTIDPKDGSITTNNIGGTGKNNINDAIGTLNQSNQELGNKITNLGDQLQQVFYDTNKRIDDVEKKANAGIAAAMALENAPFVAGKYTYAVGAAYHGGENAVGVTLRKTSDNGRWSITGGVAAASQGEPSVRVGISGVIN
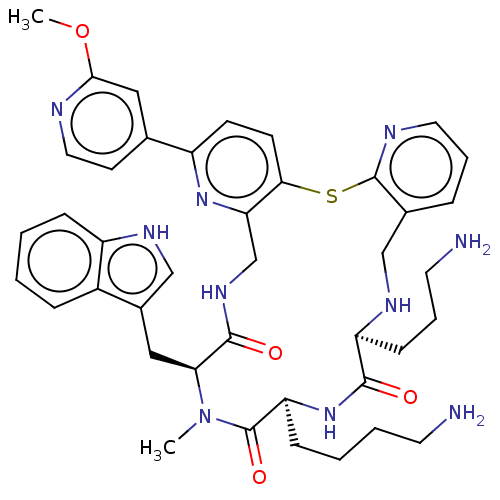
Inhibitor
Name:
BDBM281023
Synonyms:
(7S,10S,13S)-13-((1H-Indol-3-yl)methyl)-10-(4-aminobutyl)-7-(3-aminopropyl)-18-(2-methoxypyridin-4-yl)-12-methyl-6,7,9,10,12,13,15,16-octahydrodipyrido[2,3-b:2',3'-p][1,5,8,11,14]thiatetraazacycloheptadecine-8,11,14(5H)-trione | US10030047, Example 217
Type:
Small organic molecule
Emp. Form.:
C41H50N10O4S
Mol. Mass.:
778.965
SMILES:
COc1cc(ccn1)-c1ccc2Sc3ncccc3CN[C@@H](CCCN)C(=O)N[C@@H](CCCCN)C(=O)N(C)[C@@H](Cc3c[nH]c4ccccc34)C(=O)NCc2n1 |r|