Reaction Details  Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
Prothrombin
Ligand
BDBM50194743
Substrate
n/a
Meas. Tech.
ChEBML_398435
IC50
770±n/a nM
Citation
 Joossens, J; Van der Veken, P; Surpateanu, G; Lambeir, AM; El-Sayed, I; Ali, OM; Augustyns, K; Haemers, A Diphenyl phosphonate inhibitors for the urokinase-type plasminogen activator: optimization of the P4 position. J Med Chem 49:5785-93 (2006) [PubMed] Article
Joossens, J; Van der Veken, P; Surpateanu, G; Lambeir, AM; El-Sayed, I; Ali, OM; Augustyns, K; Haemers, A Diphenyl phosphonate inhibitors for the urokinase-type plasminogen activator: optimization of the P4 position. J Med Chem 49:5785-93 (2006) [PubMed] ArticleMore Info.:
Target
Name:
Prothrombin
Synonyms:
Activation peptide fragment 1 | Activation peptide fragment 2 | Coagulation factor II | F2 | Prothrombin precursor | THRB_HUMAN | Thrombin heavy chain | Thrombin light chain
Type:
Protein
Mol. Mass.:
70029.57
Organism:
Homo sapiens (Human)
Description:
P00734
Residue:
622
Sequence:
MAHVRGLQLPGCLALAALCSLVHSQHVFLAPQQARSLLQRVRRANTFLEEVRKGNLERECVEETCSYEEAFEALESSTATDVFWAKYTACETARTPRDKLAACLEGNCAEGLGTNYRGHVNITRSGIECQLWRSRYPHKPEINSTTHPGADLQENFCRNPDSSTTGPWCYTTDPTVRRQECSIPVCGQDQVTVAMTPRSEGSSVNLSPPLEQCVPDRGQQYQGRLAVTTHGLPCLAWASAQAKALSKHQDFNSAVQLVENFCRNPDGDEEGVWCYVAGKPGDFGYCDLNYCEEAVEEETGDGLDEDSDRAIEGRTATSEYQTFFNPRTFGSGEADCGLRPLFEKKSLEDKTERELLESYIDGRIVEGSDAEIGMSPWQVMLFRKSPQELLCGASLISDRWVLTAAHCLLYPPWDKNFTENDLLVRIGKHSRTRYERNIEKISMLEKIYIHPRYNWRENLDRDIALMKLKKPVAFSDYIHPVCLPDRETAASLLQAGYKGRVTGWGNLKETWTANVGKGQPSVLQVVNLPIVERPVCKDSTRIRITDNMFCAGYKPDEGKRGDACEGDSGGPFVMKSPFNNRWYQMGIVSWGEGCDRDGKYGFYTHVFRLKKWIQKVIDQFGE
Inhibitor
Name:
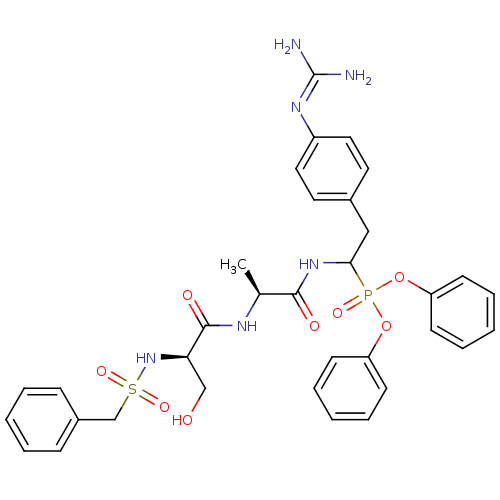
BDBM50194743
Synonyms:
CHEMBL214814 | diphenyl 1-[(N-alpha-toluenesulfonyl-D-seryl)-L-alanyl]amino-2-(4-guanidinophenyl)ethanephosphonate | {2-(4-guanidino-phenyl)-1-[(S)-2-((R)-3-hydroxy-2-phenylmethanesulfonylamino-propionylamino)-propionylamino]-ethyl}-phosphonic acid diphenyl ester
Type:
Small organic molecule
Emp. Form.:
C34H39N6O8PS
Mol. Mass.:
722.748
SMILES:
[#6]-[#6@H](-[#7]-[#6](=O)-[#6@@H](-[#6]-[#8])-[#7]S(=O)(=O)[#6]-c1ccccc1)-[#6](=O)-[#7]-[#6](-[#6]-c1ccc(cc1)\[#7]=[#6](\[#7])-[#7])P(=O)([#8]-c1ccccc1)[#8]-c1ccccc1