Reaction Details  Report a problem with these data
Report a problem with these data
 Report a problem with these data
Report a problem with these dataTarget
5-hydroxytryptamine receptor 2A
Ligand
BDBM50066912
Substrate
n/a
Meas. Tech.
ChEMBL_2661 (CHEMBL617910)
Ki
25±n/a nM
Citation
 Campiani, G; Butini, S; Gemma, S; Nacci, V; Fattorusso, C; Catalanotti, B; Giorgi, G; Cagnotto, A; Goegan, M; Mennini, T; Minetti, P; Di Cesare, MA; Mastroianni, D; Scafetta, N; Galletti, B; Stasi, MA; Castorina, M; Pacifici, L; Ghirardi, O; Tinti, O; Carminati, P Pyrrolo[1,3]benzothiazepine-based atypical antipsychotic agents. Synthesis, structure-activity relationship, molecular modeling, and biological studies. J Med Chem 45:344-59 (2002) [PubMed]
Campiani, G; Butini, S; Gemma, S; Nacci, V; Fattorusso, C; Catalanotti, B; Giorgi, G; Cagnotto, A; Goegan, M; Mennini, T; Minetti, P; Di Cesare, MA; Mastroianni, D; Scafetta, N; Galletti, B; Stasi, MA; Castorina, M; Pacifici, L; Ghirardi, O; Tinti, O; Carminati, P Pyrrolo[1,3]benzothiazepine-based atypical antipsychotic agents. Synthesis, structure-activity relationship, molecular modeling, and biological studies. J Med Chem 45:344-59 (2002) [PubMed]More Info.:
Target
Name:
5-hydroxytryptamine receptor 2A
Synonyms:
5-HT-2A | 5-HT2 | 5-HT2A | 5-hydroxytryptamine receptor 2A (5-HT2A) | 5-hydroxytryptamine receptor 2A (5HT2A) | 5HT2A_RAT | Htr2 | Htr2a | Serotonin Receptor 2A
Type:
G Protein-Coupled Receptor (GPCR)
Mol. Mass.:
52852.05
Organism:
Rattus norvegicus (rat)
Description:
Rat cortex membranes 5-HT2A receptors.
Residue:
471
Sequence:
MEILCEDNISLSSIPNSLMQLGDGPRLYHNDFNSRDANTSEASNWTIDAENRTNLSCEGYLPPTCLSILHLQEKNWSALLTTVVIILTIAGNILVIMAVSLEKKLQNATNYFLMSLAIADMLLGFLVMPVSMLTILYGYRWPLPSKLCAIWIYLDVLFSTASIMHLCAISLDRYVAIQNPIHHSRFNSRTKAFLKIIAVWTISVGISMPIPVFGLQDDSKVFKEGSCLLADDNFVLIGSFVAFFIPLTIMVITYFLTIKSLQKEATLCVSDLSTRAKLASFSFLPQSSLSSEKLFQRSIHREPGSYAGRRTMQSISNEQKACKVLGIVFFLFVVMWCPFFITNIMAVICKESCNENVIGALLNVFVWIGYLSSAVNPLVYTLFNKTYRSAFSRYIQCQYKENRKPLQLILVNTIPALAYKSSQLQVGQKKNSQEDAEQTVDDCSMVTLGKQQSEENCTDNIETVNEKVSCV
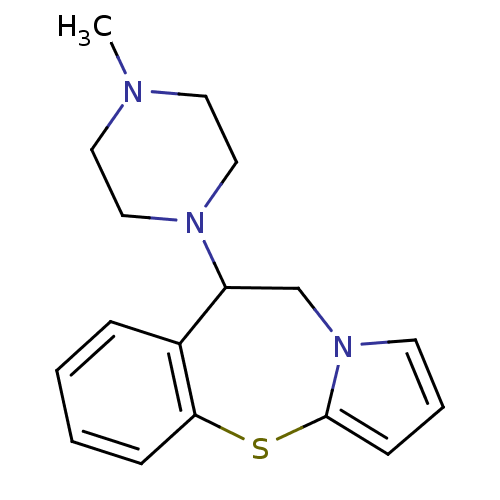
Inhibitor
Name:
BDBM50066912
Synonyms:
(+/-)9-(4-Methyl-piperazin-1-yl)-9,10-dihydro-4-thia-10a-aza-benzo[f]azulene | (R)-(-)9-(4-Methyl-piperazin-1-yl)-9,10-dihydro-4-thia-10a-aza-benzo[f]azulene | (S)-(+)9-(4-Methyl-piperazin-1-yl)-9,10-dihydro-4-thia-10a-aza-benzo[f]azulene(ST1622) | 9-(4-Methyl-piperazin-1-yl)-9,10-dihydro-4-thia-10a-aza-benzo[f]azulene | CHEMBL333246
Type:
Small organic molecule
Emp. Form.:
C17H21N3S
Mol. Mass.:
299.434
SMILES:
CN1CCN(CC1)C1Cn2cccc2Sc2ccccc12