

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM1182 CHEMBL80882::N-(3-{cyclopropyl[6-(1,3-dicyclopropylpropan-2-yl)-4-hydroxy-2-oxo-2H-pyran-3-yl]methyl}phenyl)-4-fluorobenzene-1-sulfonamide::N-[3-[Cyclopropyl[6-[2-cyclopropyl-1-(cyclopropyl-methyl)ethyl]-4-hydroxy-2-oxo-2H-pyran-3-yl]methyl]phenyl]-4-flurobenzenesulfonamide::Sulfonamide-Containing 4-Hydroxy-2-pyrone deriv. XXII
SMILES: Oc1cc(oc(=O)c1C(C1CC1)c1cccc(NS(=O)(=O)c2ccc(F)cc2)c1)C(CC1CC1)CC1CC1
InChI Key: InChIKey=SLZNWUVSDZUVTO-UHFFFAOYSA-N
Data: 2 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Human immunodeficiency virus type 1 protease (Human immunodeficiency virus type 1) | BDBM1182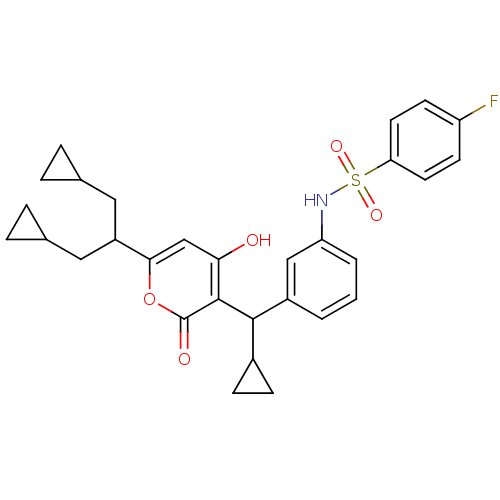 (CHEMBL80882 | N-(3-{cyclopropyl[6-(1,3-dicycloprop...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | 16 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of HIV-1 protease. | Bioorg Med Chem Lett 7: 399-402 (1997) Article DOI: 10.1016/S0960-894X(97)00031-0 BindingDB Entry DOI: 10.7270/Q21R6QH6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| HIV-1 Protease (Human immunodeficiency virus type 1) | BDBM1182 (CHEMBL80882 | N-(3-{cyclopropyl[6-(1,3-dicycloprop...) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 16 | -10.5 | n/a | n/a | n/a | n/a | n/a | 5.0 | 22 |
Upjohn | Assay Description HIV-1 protease was purified and refolded from E. coli inclusion bodies. The substrate used spans the p17-p24 processing site (R-V-S-Q-N-Y-P-I-V-Q-N-K... | J Med Chem 39: 2400-10 (1996) Article DOI: 10.1021/jm950888f BindingDB Entry DOI: 10.7270/Q20K26RK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||