

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM12597 CHEMBL48046::RPR128515::methyl (2R,3R)-2-{3-[amino(imino)methyl]benzyl}-3-({[3-(aminomethyl)-1,1-biphenyl-4-yl]carbonyl}amino)butanoate::methyl (2R,3R)-3-({4-[3-(aminomethyl)phenyl]phenyl}formamido)-2-[(3-carbamimidoylphenyl)methyl]butanoate
SMILES: COC(=O)[C@H](Cc1cccc(c1)C(N)=N)[C@@H](C)NC(=O)c1ccc(cc1)-c1cccc(CN)c1
InChI Key: InChIKey=XFKVLKLCLYJKNF-MZNJEOGPSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Coagulation factor X (Homo sapiens (Human)) | BDBM12597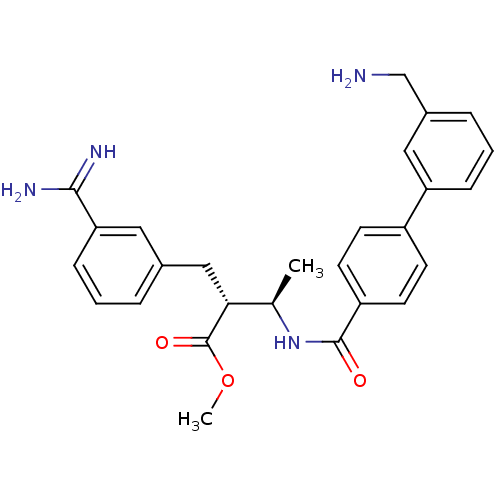 (CHEMBL48046 | RPR128515 | methyl (2R,3R)-2-{3-[ami...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.900 | -12.2 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
Aventis Pharma | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | J Med Chem 43: 3226-32 (2000) Article DOI: 10.1021/jm000940u BindingDB Entry DOI: 10.7270/Q25H7DHP | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM12597 (CHEMBL48046 | RPR128515 | methyl (2R,3R)-2-{3-[ami...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 0.900 | -12.2 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
Aventis Pharma | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | J Med Chem 46: 685-90 (2003) Article DOI: 10.1021/jm0203837 BindingDB Entry DOI: 10.7270/Q2VH5M2S | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM12597 (CHEMBL48046 | RPR128515 | methyl (2R,3R)-2-{3-[ami...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Similars | PDB PubMed | 0.900 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals Curated by ChEMBL | Assay Description In vitro inhibitory activity against human Coagulation factor Xa | Bioorg Med Chem Lett 12: 1667-70 (2002) BindingDB Entry DOI: 10.7270/Q2GM86MR | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Trypsin-1 (Homo sapiens (Human)) | BDBM12597 (CHEMBL48046 | RPR128515 | methyl (2R,3R)-2-{3-[ami...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Similars | PubMed | 69 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Aventis Pharmaceuticals Curated by ChEMBL | Assay Description In vitro inhibitory activity against human trypsin | Bioorg Med Chem Lett 12: 1667-70 (2002) BindingDB Entry DOI: 10.7270/Q2GM86MR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Trypsin (Bos taurus (bovine)) | BDBM12597 (CHEMBL48046 | RPR128515 | methyl (2R,3R)-2-{3-[ami...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Similars | DrugBank MMDB PDB Article PubMed | 69 | -9.66 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
Aventis Pharma | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | J Med Chem 46: 685-90 (2003) Article DOI: 10.1021/jm0203837 BindingDB Entry DOI: 10.7270/Q2VH5M2S | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Trypsin (Bos taurus (bovine)) | BDBM12597 (CHEMBL48046 | RPR128515 | methyl (2R,3R)-2-{3-[ami...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Similars | DrugBank MMDB PDB Article PubMed | 69 | -9.66 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
Aventis Pharma | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | J Med Chem 43: 3226-32 (2000) Article DOI: 10.1021/jm000940u BindingDB Entry DOI: 10.7270/Q25H7DHP | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM12597 (CHEMBL48046 | RPR128515 | methyl (2R,3R)-2-{3-[ami...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | >3.00E+3 | >-7.45 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
Aventis Pharma | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | J Med Chem 46: 685-90 (2003) Article DOI: 10.1021/jm0203837 BindingDB Entry DOI: 10.7270/Q2VH5M2S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM12597 (CHEMBL48046 | RPR128515 | methyl (2R,3R)-2-{3-[ami...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL DrugBank MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | >3.00E+3 | >-7.45 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
Aventis Pharma | Assay Description The enzyme reactions were initiated by the addition of substrate, and the color developed from the release of p-nitroanilide from each chromogenic su... | J Med Chem 43: 3226-32 (2000) Article DOI: 10.1021/jm000940u BindingDB Entry DOI: 10.7270/Q25H7DHP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||