

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM13606 (3S)-3-{[(1S)-1-{[(1S)-1-carbamoylpentyl]carbamoyl}-2-[4-(sulfooxy)phenyl]ethyl]carbamoyl}-3-acetamidopropanoic acid::CHEMBL175257::Compound I::N-acetyl-Asp-Tyr(SO3H)-Nle-NH2::sulfotyrosyl tripeptide I
SMILES: CCCC[C@H](NC(=O)[C@H](Cc1ccc(OS(O)(=O)=O)cc1)NC(=O)[C@H](CC(O)=O)NC(C)=O)C(N)=O
InChI Key: InChIKey=VYEUAVNMANWCSE-ULQDDVLXSA-N
Data: 2 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Protein-tyrosine phosphatase 1B (Homo sapiens (Human)) | BDBM13606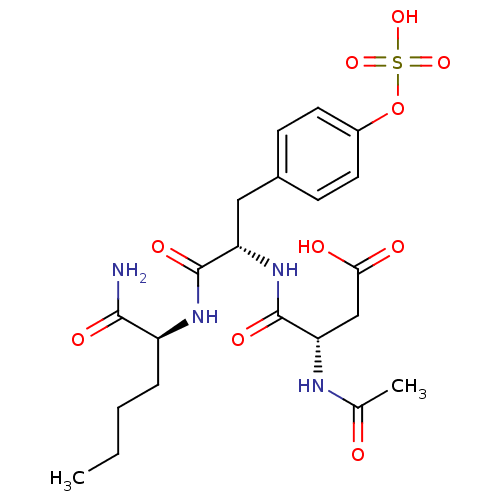 ((3S)-3-{[(1S)-1-{[(1S)-1-carbamoylpentyl]carbamoyl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | PubMed | 5.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pharmacia Corporation Curated by ChEMBL | Assay Description Inhibitory activity against human protein tyrosine phosphatase 1B (PTP1B) | Bioorg Med Chem Lett 13: 971-5 (2003) BindingDB Entry DOI: 10.7270/Q24M93WC | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protein-Tyrosine Phosphatase 1B (PTP1B) (Rattus norvegicus (rat)) | BDBM13606 ((3S)-3-{[(1S)-1-{[(1S)-1-carbamoylpentyl]carbamoyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL KEGG PC cid PC sid UniChem Similars | Article PubMed | 5.00E+3 | -7.15 | n/a | n/a | n/a | n/a | n/a | 7.2 | 22 |
Pharmacia Corporation | Assay Description Hydrolysis of substrate p-nitrophenyl phosphate was monitored on a 96-well microplate reader. Concentrations of PTP1B inhibitors that resulted in 50%... | Biochemistry 40: 5642-54 (2001) Article DOI: 10.1021/bi002865v BindingDB Entry DOI: 10.7270/Q2D50K6S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||