 Found 5 hits for monomerid = 13966
Found 5 hits for monomerid = 13966 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Tyrosine-protein phosphatase non-receptor type 2
(Homo sapiens (Human)) | BDBM13966
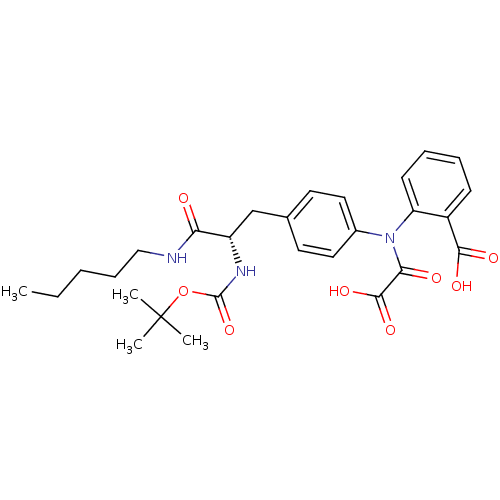
(2-({4-[(2S)-2-{[(tert-butoxy)carbonyl]amino}-2-(pe...)Show SMILES CCCCCNC(=O)[C@H](Cc1ccc(cc1)N(C(=O)C(O)=O)c1ccccc1C(O)=O)NC(=O)OC(C)(C)C |r| Show InChI InChI=1S/C28H35N3O8/c1-5-6-9-16-29-23(32)21(30-27(38)39-28(2,3)4)17-18-12-14-19(15-13-18)31(24(33)26(36)37)22-11-8-7-10-20(22)25(34)35/h7-8,10-15,21H,5-6,9,16-17H2,1-4H3,(H,29,32)(H,30,38)(H,34,35)(H,36,37)/t21-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.30E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
| Assay Description
The phosphatase activity resulted in the formation of the colored product p-nitrophenol, which was continuously monitored at 405 nm every 30 s for 15... |
J Med Chem 46: 2093-103 (2003)
Article DOI: 10.1021/jm0205696
BindingDB Entry DOI: 10.7270/Q26H4FPH |
More data for this
Ligand-Target Pair | |
Protein-tyrosine phosphatase 1B
(Homo sapiens (Human)) | BDBM13966

(2-({4-[(2S)-2-{[(tert-butoxy)carbonyl]amino}-2-(pe...)Show SMILES CCCCCNC(=O)[C@H](Cc1ccc(cc1)N(C(=O)C(O)=O)c1ccccc1C(O)=O)NC(=O)OC(C)(C)C |r| Show InChI InChI=1S/C28H35N3O8/c1-5-6-9-16-29-23(32)21(30-27(38)39-28(2,3)4)17-18-12-14-19(15-13-18)31(24(33)26(36)37)22-11-8-7-10-20(22)25(34)35/h7-8,10-15,21H,5-6,9,16-17H2,1-4H3,(H,29,32)(H,30,38)(H,34,35)(H,36,37)/t21-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.58E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Abbott Laboratories
Curated by ChEMBL
| Assay Description
Inhibition of PTP1B |
J Med Chem 52: 3159-65 (2009)
Article DOI: 10.1021/jm801444x
BindingDB Entry DOI: 10.7270/Q2FF3TM8 |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Protein-tyrosine phosphatase 1B
(Homo sapiens (Human)) | BDBM13966

(2-({4-[(2S)-2-{[(tert-butoxy)carbonyl]amino}-2-(pe...)Show SMILES CCCCCNC(=O)[C@H](Cc1ccc(cc1)N(C(=O)C(O)=O)c1ccccc1C(O)=O)NC(=O)OC(C)(C)C |r| Show InChI InChI=1S/C28H35N3O8/c1-5-6-9-16-29-23(32)21(30-27(38)39-28(2,3)4)17-18-12-14-19(15-13-18)31(24(33)26(36)37)22-11-8-7-10-20(22)25(34)35/h7-8,10-15,21H,5-6,9,16-17H2,1-4H3,(H,29,32)(H,30,38)(H,34,35)(H,36,37)/t21-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
| 1.73E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of PTP1B (unknown origin) |
Citation and Details
Article DOI: 10.1007/s00044-010-9365-7
BindingDB Entry DOI: 10.7270/Q25D8VRJ |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Protein-Tyrosine Phosphatase 1B (PTP1B)
(Homo sapiens (Human)) | BDBM13966

(2-({4-[(2S)-2-{[(tert-butoxy)carbonyl]amino}-2-(pe...)Show SMILES CCCCCNC(=O)[C@H](Cc1ccc(cc1)N(C(=O)C(O)=O)c1ccccc1C(O)=O)NC(=O)OC(C)(C)C |r| Show InChI InChI=1S/C28H35N3O8/c1-5-6-9-16-29-23(32)21(30-27(38)39-28(2,3)4)17-18-12-14-19(15-13-18)31(24(33)26(36)37)22-11-8-7-10-20(22)25(34)35/h7-8,10-15,21H,5-6,9,16-17H2,1-4H3,(H,29,32)(H,30,38)(H,34,35)(H,36,37)/t21-/m0/s1 | PDB
MMDB
B.MOAD
DrugBank
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.73E+4 | -6.43 | n/a | n/a | n/a | n/a | n/a | 7.5 | 22 |
Abbott Laboratories
| Assay Description
The phosphatase activity resulted in the formation of the colored product p-nitrophenol, which was continuously monitored at 405 nm every 30 s for 15... |
J Med Chem 46: 2093-103 (2003)
Article DOI: 10.1021/jm0205696
BindingDB Entry DOI: 10.7270/Q26H4FPH |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |
Protein-tyrosine phosphatase 1B
(Homo sapiens (Human)) | BDBM13966

(2-({4-[(2S)-2-{[(tert-butoxy)carbonyl]amino}-2-(pe...)Show SMILES CCCCCNC(=O)[C@H](Cc1ccc(cc1)N(C(=O)C(O)=O)c1ccccc1C(O)=O)NC(=O)OC(C)(C)C |r| Show InChI InChI=1S/C28H35N3O8/c1-5-6-9-16-29-23(32)21(30-27(38)39-28(2,3)4)17-18-12-14-19(15-13-18)31(24(33)26(36)37)22-11-8-7-10-20(22)25(34)35/h7-8,10-15,21H,5-6,9,16-17H2,1-4H3,(H,29,32)(H,30,38)(H,34,35)(H,36,37)/t21-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
Similars
| Article
| 1.74E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA
| Assay Description
Inhibition of PTP1B (unknown origin) |
Citation and Details
Article DOI: 10.1007/s00044-010-9365-7
BindingDB Entry DOI: 10.7270/Q25D8VRJ |
More data for this
Ligand-Target Pair | 
3D Structure (docked) |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data