

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM14308 (2S)-N-[3-(2-amino-1H-1,3-benzodiazol-5-yl)-1-(1,3-benzothiazol-2-yl)-1-oxopropan-2-yl]-3-phenyl-2-[(2R)-3-phenyl-2-(phenylmethane)sulfonamidopropanamido]propanamide::2-Aminobenzimidazole Compound 12
SMILES: Nc1nc2ccc(CC(NC(=O)[C@H](Cc3ccccc3)NC(=O)[C@@H](Cc3ccccc3)NS(=O)(=O)Cc3ccccc3)C(=O)c3nc4ccccc4s3)cc2[nH]1
InChI Key: InChIKey=OYCGDTJZFBPAEP-CIDCSGNASA-N
Data: 1 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Tryptase (Homo sapiens (Human)) | BDBM14308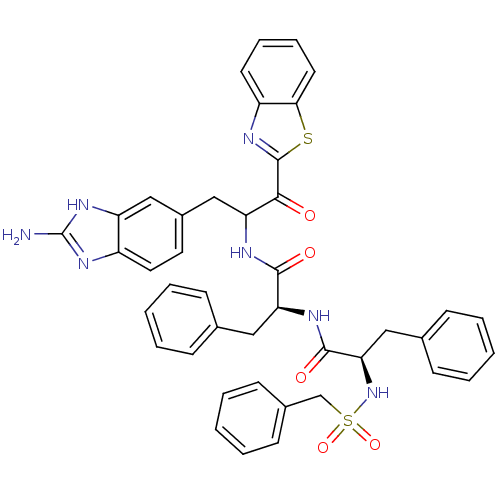 ((2S)-N-[3-(2-amino-1H-1,3-benzodiazol-5-yl)-1-(1,3...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem Similars | Article PubMed | 1.80E+4 | -6.40 | n/a | n/a | n/a | n/a | n/a | 7.4 | 22 |
Celera | Assay Description Each enzyme was assayed with a set of different concentrations of each inhibitor. After addition of the appropriate substrate, the rate of hydrolysis... | Biochemistry 45: 5964-73 (2006) Article DOI: 10.1021/bi060173m BindingDB Entry DOI: 10.7270/Q2W09450 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||