

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM196 A-74704::CHEMBL307193::benzyl N-[(1S)-1-{[(2S,4S)-4-[(2S)-2-{[(benzyloxy)carbonyl]amino}-3-methylbutanamido]-3-hydroxy-1,5-diphenylpentan-2-yl]carbamoyl}-2-methylpropyl]carbamate
SMILES: CC(C)[C@H](NC(=O)OCc1ccccc1)C(=O)N[C@@H](Cc1ccccc1)C(O)[C@H](Cc1ccccc1)NC(=O)[C@@H](NC(=O)OCc1ccccc1)C(C)C
InChI Key: InChIKey=GEANBHANAKKWSL-ZQWQDMLBSA-N
PDB links: 1 PDB ID matches this monomer. 7 PDB IDs contain inhibitors having a similarity of 90% to this monomer.
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| HIV-1 Protease (Human immunodeficiency virus type 1) | BDBM196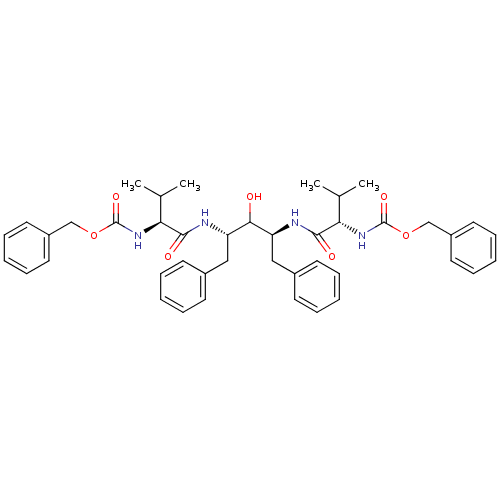 (A-74704 | CHEMBL307193 | benzyl N-[(1S)-1-{[(2S,4S...) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB Article PubMed | 4.5 | -11.6 | n/a | n/a | n/a | n/a | n/a | 4.7 | 30 |
Abbott Laboratories | Assay Description HIV-1 protease activity was measured by a continuous fluorometric assay using the internally quenched fluorogenic substrate DABCYL-GABA-Ser-Gln-Tyr-P... | Science 249: 527-33 (1990) Article DOI: 10.1126/science.2200122 BindingDB Entry DOI: 10.7270/Q2QC01NV | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Human immunodeficiency virus type 1 protease (Human immunodeficiency virus type 1) | BDBM196 (A-74704 | CHEMBL307193 | benzyl N-[(1S)-1-{[(2S,4S...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PubMed | n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp and Dohme Research Laboratories Curated by ChEMBL | Assay Description The compound was tested for its affinity against HIV-1 protease | J Med Chem 34: 2305-14 (1991) BindingDB Entry DOI: 10.7270/Q2862H21 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Human immunodeficiency virus type 1 protease (Human immunodeficiency virus type 1) | BDBM196 (A-74704 | CHEMBL307193 | benzyl N-[(1S)-1-{[(2S,4S...) | PDB MMDB UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Patents Similars | MMDB PubMed | n/a | n/a | 3 | n/a | n/a | n/a | n/a | n/a | n/a |
Université de la Méditerranée Curated by ChEMBL | Assay Description Inhibitory activity of the compound was evaluated against HIV-protease was evaluated | Bioorg Med Chem Lett 8: 1861-6 (1999) BindingDB Entry DOI: 10.7270/Q25T3JNQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||