 Found 10 hits for monomerid = 205419
Found 10 hits for monomerid = 205419 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Protein O-GlcNAcase
(Homo sapiens (Human)) | BDBM205419
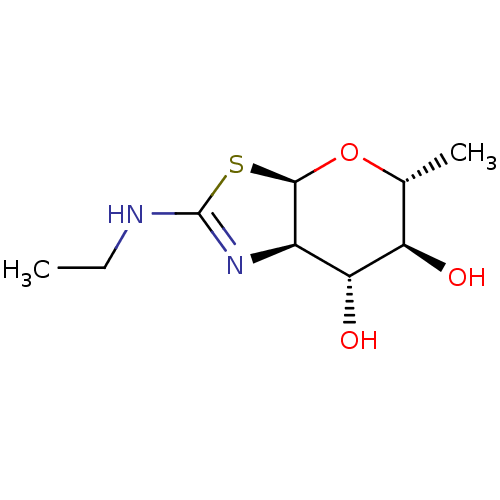
(US9243020, 1 | US9815861, Example 1)Show SMILES CCNC1=N[C@H]2[C@H](O[C@H](C)[C@@H](O)[C@@H]2O)S1 |r,t:3| Show InChI InChI=1S/C9H16N2O3S/c1-3-10-9-11-5-7(13)6(12)4(2)14-8(5)15-9/h4-8,12-13H,3H2,1-2H3,(H,10,11)/t4-,5-,6-,7-,8-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| 69 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co.
Curated by ChEMBL
| Assay Description
Inhibition of recombinant human OGA |
J Med Chem 62: 10062-10097 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01090 |
More data for this
Ligand-Target Pair | |
Protein O-GlcNAcase
(Homo sapiens (Human)) | BDBM205419

(US9243020, 1 | US9815861, Example 1)Show SMILES CCNC1=N[C@H]2[C@H](O[C@H](C)[C@@H](O)[C@@H]2O)S1 |r,t:3| Show InChI InChI=1S/C9H16N2O3S/c1-3-10-9-11-5-7(13)6(12)4(2)14-8(5)15-9/h4-8,12-13H,3H2,1-2H3,(H,10,11)/t4-,5-,6-,7-,8-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 70 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Alectos Therapeutics, Inc.; Merck Sharp & Dohme Corp.
US Patent
| Assay Description
Enzymatic reactions were carried out in a reaction containing 50 mM NaH2PO4, 100 mM NaCl and 0.1% BSA (pH 7.0) using 2 mM 4-Methylumbelliferyl N-acet... |
US Patent US9815861 (2017)
BindingDB Entry DOI: 10.7270/Q2R78HJ8 |
More data for this
Ligand-Target Pair | |
Protein O-GlcNAcase
(Homo sapiens (Human)) | BDBM205419

(US9243020, 1 | US9815861, Example 1)Show SMILES CCNC1=N[C@H]2[C@H](O[C@H](C)[C@@H](O)[C@@H]2O)S1 |r,t:3| Show InChI InChI=1S/C9H16N2O3S/c1-3-10-9-11-5-7(13)6(12)4(2)14-8(5)15-9/h4-8,12-13H,3H2,1-2H3,(H,10,11)/t4-,5-,6-,7-,8-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| US Patent
| 70 | -9.75 | n/a | n/a | n/a | n/a | n/a | 7.0 | 25 |
Alectos Therapeutics Inc.; Merck Sharp & Dohme Corp.
US Patent
| Assay Description
Enzymatic reactions were carried out in a reaction containing 50 mM NaH2PO4, 100 mM NaCl and 0.1% BSA (pH 7.0) using 2 mM 4-Methylumbelliferyl N-acet... |
US Patent US9243020 (2016)
BindingDB Entry DOI: 10.7270/Q2542MD4 |
More data for this
Ligand-Target Pair | |
Voltage-dependent L-type calcium channel subunit alpha-1C
(Homo sapiens (Human)) | BDBM205419

(US9243020, 1 | US9815861, Example 1)Show SMILES CCNC1=N[C@H]2[C@H](O[C@H](C)[C@@H](O)[C@@H]2O)S1 |r,t:3| Show InChI InChI=1S/C9H16N2O3S/c1-3-10-9-11-5-7(13)6(12)4(2)14-8(5)15-9/h4-8,12-13H,3H2,1-2H3,(H,10,11)/t4-,5-,6-,7-,8-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co.
Curated by ChEMBL
| Assay Description
Inhibition of Cav1.2 (unknown origin) |
J Med Chem 62: 10062-10097 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01090 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM205419

(US9243020, 1 | US9815861, Example 1)Show SMILES CCNC1=N[C@H]2[C@H](O[C@H](C)[C@@H](O)[C@@H]2O)S1 |r,t:3| Show InChI InChI=1S/C9H16N2O3S/c1-3-10-9-11-5-7(13)6(12)4(2)14-8(5)15-9/h4-8,12-13H,3H2,1-2H3,(H,10,11)/t4-,5-,6-,7-,8-/m1/s1 | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co.
Curated by ChEMBL
| Assay Description
Inhibition of CYP2D6 (unknown origin) |
J Med Chem 62: 10062-10097 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01090 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM205419

(US9243020, 1 | US9815861, Example 1)Show SMILES CCNC1=N[C@H]2[C@H](O[C@H](C)[C@@H](O)[C@@H]2O)S1 |r,t:3| Show InChI InChI=1S/C9H16N2O3S/c1-3-10-9-11-5-7(13)6(12)4(2)14-8(5)15-9/h4-8,12-13H,3H2,1-2H3,(H,10,11)/t4-,5-,6-,7-,8-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co.
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 (unknown origin) |
J Med Chem 62: 10062-10097 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01090 |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM205419

(US9243020, 1 | US9815861, Example 1)Show SMILES CCNC1=N[C@H]2[C@H](O[C@H](C)[C@@H](O)[C@@H]2O)S1 |r,t:3| Show InChI InChI=1S/C9H16N2O3S/c1-3-10-9-11-5-7(13)6(12)4(2)14-8(5)15-9/h4-8,12-13H,3H2,1-2H3,(H,10,11)/t4-,5-,6-,7-,8-/m1/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co.
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 (unknown origin) |
J Med Chem 62: 10062-10097 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01090 |
More data for this
Ligand-Target Pair | |
1,3-beta-glucan synthase component GSC2
(Saccharomyces cerevisiae) | BDBM205419

(US9243020, 1 | US9815861, Example 1)Show SMILES CCNC1=N[C@H]2[C@H](O[C@H](C)[C@@H](O)[C@@H]2O)S1 |r,t:3| Show InChI InChI=1S/C9H16N2O3S/c1-3-10-9-11-5-7(13)6(12)4(2)14-8(5)15-9/h4-8,12-13H,3H2,1-2H3,(H,10,11)/t4-,5-,6-,7-,8-/m1/s1 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co.
Curated by ChEMBL
| Assay Description
Displacement of MK-0499 from human ERG |
J Med Chem 62: 10062-10097 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01090 |
More data for this
Ligand-Target Pair | |
Nuclear receptor subfamily 1 group I member 2
(Homo sapiens (Human)) | BDBM205419

(US9243020, 1 | US9815861, Example 1)Show SMILES CCNC1=N[C@H]2[C@H](O[C@H](C)[C@@H](O)[C@@H]2O)S1 |r,t:3| Show InChI InChI=1S/C9H16N2O3S/c1-3-10-9-11-5-7(13)6(12)4(2)14-8(5)15-9/h4-8,12-13H,3H2,1-2H3,(H,10,11)/t4-,5-,6-,7-,8-/m1/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a |
Merck & Co.
Curated by ChEMBL
| Assay Description
Activation of PXR (unknown origin) assessed as CYP3A4 induction |
J Med Chem 62: 10062-10097 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01090 |
More data for this
Ligand-Target Pair | |
Sodium channel protein type 5 subunit alpha
(Homo sapiens (Human)) | BDBM205419

(US9243020, 1 | US9815861, Example 1)Show SMILES CCNC1=N[C@H]2[C@H](O[C@H](C)[C@@H](O)[C@@H]2O)S1 |r,t:3| Show InChI InChI=1S/C9H16N2O3S/c1-3-10-9-11-5-7(13)6(12)4(2)14-8(5)15-9/h4-8,12-13H,3H2,1-2H3,(H,10,11)/t4-,5-,6-,7-,8-/m1/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| PC cid
PC sid
UniChem
| Article
PubMed
| n/a | n/a | >3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co.
Curated by ChEMBL
| Assay Description
Inhibition of Nav1.5 (unknown origin) |
J Med Chem 62: 10062-10097 (2019)
Article DOI: 10.1021/acs.jmedchem.9b01090 |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data