

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM23207 (1R,2R,5S,8R,9R,10R,13R,14R,17S,19R)-5-(hydroxymethyl)-1,2,14,18,18-pentamethyl-8-(prop-1-en-2-yl)pentacyclo[11.8.0.0^{2,10}.0^{5,9}.0^{14,19}]henicosan-17-ol::Betulin, 23::Betulinol
SMILES: [H][C@]12[C@@H](CC[C@]1(CO)CC[C@]1(C)[C@]2([H])CC[C@]2([H])[C@@]3(C)CC[C@H](O)C(C)(C)[C@]3([H])CC[C@@]12C)C(C)=C
InChI Key: InChIKey=FVWJYYTZTCVBKE-ROUWMTJPSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Glycogen phosphorylase, muscle form (Oryctolagus cuniculus (rabbit)) | BDBM23207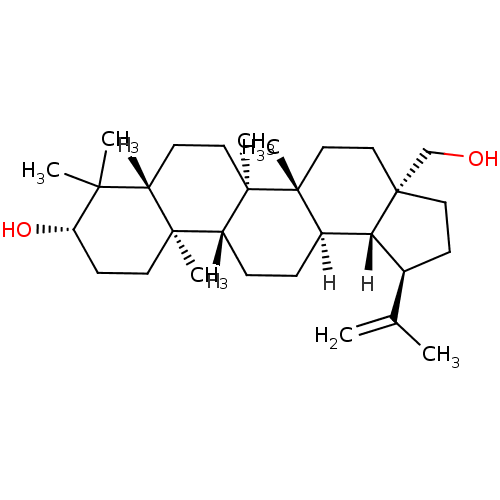 ((1R,2R,5S,8R,9R,10R,13R,14R,17S,19R)-5-(hydroxymet...) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.70E+4 | n/a | n/a | n/a | n/a | 7.2 | 22 |
China Pharmaceutical University | Assay Description The activity of the compounds is determined by measuring the inhibitory effect of the compounds in the direction of glycogen synthesis, the conversio... | J Med Chem 51: 3540-54 (2008) Article DOI: 10.1021/jm8000949 BindingDB Entry DOI: 10.7270/Q2WQ0233 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| PKC alpha and beta-2 (Homo sapiens (Human)) | BDBM23207 ((1R,2R,5S,8R,9R,10R,13R,14R,17S,19R)-5-(hydroxymet...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of PKC beta2 | J Nat Prod 57: 243-247 (1994) Article DOI: 10.1021/np50104a008 BindingDB Entry DOI: 10.7270/Q2TT4R0Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protein kinase C, epsilon (Homo sapiens (Human)) | BDBM23207 ((1R,2R,5S,8R,9R,10R,13R,14R,17S,19R)-5-(hydroxymet...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of PKC epsilon | J Nat Prod 57: 243-247 (1994) Article DOI: 10.1021/np50104a008 BindingDB Entry DOI: 10.7270/Q2TT4R0Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glycogen phosphorylase, muscle form (Oryctolagus cuniculus (rabbit)) | BDBM23207 ((1R,2R,5S,8R,9R,10R,13R,14R,17S,19R)-5-(hydroxymet...) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 4.15E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of rabbit muscle glycogen phosphorylase a | Bioorg Med Chem Lett 21: 338-41 (2010) Article DOI: 10.1016/j.bmcl.2010.11.004 BindingDB Entry DOI: 10.7270/Q2MC909F | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Reverse transcriptase (Human immunodeficiency virus 1) | BDBM23207 ((1R,2R,5S,8R,9R,10R,13R,14R,17S,19R)-5-(hydroxymet...) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >4.51E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Illinois Curated by ChEMBL | Assay Description Inhibition of HIV1 RT | J Nat Prod 54: 143-54 Article DOI: 10.1021/np50073a012 BindingDB Entry DOI: 10.7270/Q2NK3HTG | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Topoisomerase II alpha (HuTopoIIα) (Homo sapiens (Human)) | BDBM23207 ((1R,2R,5S,8R,9R,10R,13R,14R,17S,19R)-5-(hydroxymet...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | 3.86E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Osaka University of Pharmaceutical Sciences Curated by ChEMBL | Assay Description Inhibition of human 1 unit topoisomerase 2alpha catalytic activity assessed as relaxation of 198 ng supercoiled pBR322 DNA by agarose gel electrophor... | J Nat Prod 64: 1545-7 (2001) BindingDB Entry DOI: 10.7270/Q2X92C63 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| G-protein coupled bile acid receptor 1 (Homo sapiens (Human)) | BDBM23207 ((1R,2R,5S,8R,9R,10R,13R,14R,17S,19R)-5-(hydroxymet...) | PDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a |
Universite Louis Pasteur Curated by ChEMBL | Assay Description Agonist activity at TGR5 expressed in CHO cells by CRE-driven luciferase reporter gene assay | J Med Chem 53: 178-90 (2010) Article DOI: 10.1021/jm900872z BindingDB Entry DOI: 10.7270/Q2RJ4KDW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Pancreatic alpha-amylase (Sus scrofa (Pig)) | BDBM23207 ((1R,2R,5S,8R,9R,10R,13R,14R,17S,19R)-5-(hydroxymet...) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
School of Life Science and Biotechnology, Dalian University of Technology, Dalian 116024, Liaoning, China. Curated by ChEMBL | Assay Description Inhibition of porcine pancreatic alpha-amylase using starch as substrate preincubated for 15 mins followed by substrate addition measured after 10 mi... | Bioorg Med Chem Lett 27: 5065-5070 (2017) Article DOI: 10.1016/j.bmcl.2017.09.027 BindingDB Entry DOI: 10.7270/Q2Q81GJ7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nuclear factor NF-kappa-B (Homo sapiens (Human)) | BDBM23207 ((1R,2R,5S,8R,9R,10R,13R,14R,17S,19R)-5-(hydroxymet...) | PDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 8.80E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
The Ohio State University Curated by ChEMBL | Assay Description Inhibition of biotinylated consensus sequence binding to NF-kB p65 in human HeLa nuclear extracts after 3 hrs by ELISA | Bioorg Med Chem 26: 4452-4460 (2018) Article DOI: 10.1016/j.bmc.2018.07.025 BindingDB Entry DOI: 10.7270/Q2N300MM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Glycogen phosphorylase, muscle form (Oryctolagus cuniculus (rabbit)) | BDBM23207 ((1R,2R,5S,8R,9R,10R,13R,14R,17S,19R)-5-(hydroxymet...) | PDB MMDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | Purchase KEGG PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 1.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Chinese Academy of Sciences Curated by ChEMBL | Assay Description Inhibition of rabbit muscle glycogen phosphorylase A assessed as release of phosphate from glucose-1-phosphate after 25 mins by microplate reader bas... | Eur J Med Chem 46: 2011-21 (2011) Article DOI: 10.1016/j.ejmech.2011.02.053 BindingDB Entry DOI: 10.7270/Q2DB826B | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||