

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM26126 (3-phenylphenyl)boranediol::Phenylboronic Acid, 5
SMILES: OB(O)c1cccc(c1)-c1ccccc1
InChI Key: InChIKey=GOXICVKOZJFRMB-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Beta-lactamase (TEM-1) (Escherichia coli) | BDBM26126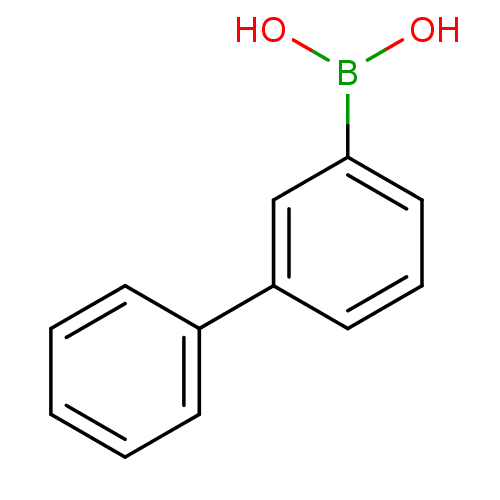 ((3-phenylphenyl)boranediol | Phenylboronic Acid, 5) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents | Article | 1.39E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibitory constant was determined against class A RTEM-1 Beta-lactamase from Escherichia coli | Bioorg Med Chem Lett 4: 1229-1234 (1994) Article DOI: 10.1016/S0960-894X(01)80336-X BindingDB Entry DOI: 10.7270/Q2W9595N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Subtilisin (Bacillus lentus) | BDBM26126 ((3-phenylphenyl)boranediol | Phenylboronic Acid, 5) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents | Article | 3.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Competitive inhibition of Subtilisin BL wild type enzyme from B. lentus | Bioorg Med Chem Lett 6: 2501-2506 (1996) Article DOI: 10.1016/0960-894X(96)00466-0 BindingDB Entry DOI: 10.7270/Q2X34XFJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Subtilisin (Bacillus licheniformis) | BDBM26126 ((3-phenylphenyl)boranediol | Phenylboronic Acid, 5) | PDB MMDB B.MOAD DrugBank GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents | Article | 3.70E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Competitive inhibition of Subtilisin Carlsberg enzyme from B. lentus | Bioorg Med Chem Lett 6: 2501-2506 (1996) Article DOI: 10.1016/0960-894X(96)00466-0 BindingDB Entry DOI: 10.7270/Q2X34XFJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Subtilisin (Bacillus lentus) | BDBM26126 ((3-phenylphenyl)boranediol | Phenylboronic Acid, 5) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents | Article | 6.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Competitive inhibition of Subtilisin BL M222S-mutated enzyme from B. lentus | Bioorg Med Chem Lett 6: 2501-2506 (1996) Article DOI: 10.1016/0960-894X(96)00466-0 BindingDB Entry DOI: 10.7270/Q2X34XFJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Subtilisin (Bacillus lentus) | BDBM26126 ((3-phenylphenyl)boranediol | Phenylboronic Acid, 5) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents | Article | 3.27E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Competitive inhibition of Subtilisin BL M222S-mutated enzyme from B. lentus | Bioorg Med Chem Lett 6: 2501-2506 (1996) Article DOI: 10.1016/0960-894X(96)00466-0 BindingDB Entry DOI: 10.7270/Q2X34XFJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Monoglyceride lipase (Homo sapiens (Human)) | BDBM26126 ((3-phenylphenyl)boranediol | Phenylboronic Acid, 5) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 7.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Oxford | Assay Description The endpoint enzymatic assay was developed to quantify human recombinant MGL activity with 2-AG. The formation of arachidonic acid and depletion of ... | J Med Chem 51: 7057-60 (2008) Article DOI: 10.1021/jm801051t BindingDB Entry DOI: 10.7270/Q25D8Q58 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Fatty-acid amide hydrolase 1 (aa 30-579) (Rattus norvegicus (rat)) | BDBM26126 ((3-phenylphenyl)boranediol | Phenylboronic Acid, 5) | PDB MMDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase PC cid PC sid UniChem Patents | Article PubMed | n/a | n/a | 130 | n/a | n/a | n/a | n/a | 7.4 | 37 |
University of Oxford | Assay Description [3H]Ethanolamine produced from [3H]AEA hydrolysis was used to calculate FAAH activity and was measured by scintillation counting of the aqueous phase... | J Med Chem 51: 7057-60 (2008) Article DOI: 10.1021/jm801051t BindingDB Entry DOI: 10.7270/Q25D8Q58 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||