

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM330060 (1R,3S,4R,12aR)-7-hydroxy-3-methyl-6,8-dioxo-N-(2,4,6-trifluorobenzyl)-1,2,3,4,6,8,12,12a-octahydro-1,4-methanodipyrido[1,2-a:1′,2′-d]pyrazine-9-carboxamide::US10689399, Compound 99::US9663528, 99::US9732092, Compound 99
SMILES: C[C@H]1C[C@@H]2C[C@H]1N1[C@H]2Cn2cc(C(=O)NCc3c(F)cc(F)cc3F)c(=O)c(O)c2C1=O
InChI Key: InChIKey=GXAXTRZNCYOPOM-DRKCQWHGSA-N
Data: 3 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Solute carrier family 22 member 2 (Homo sapiens (Human)) | BDBM330060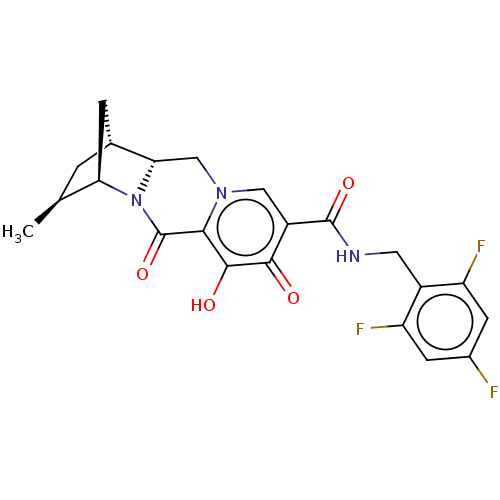 ((1R,3S,4R,12aR)-7-hydroxy-3-methyl-6,8-dioxo-N-(2,...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 1.51E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Gilead Sciences, Inc. US Patent | Assay Description The dose dependent inhibition of OCT2 mediated uptake of a model substrate 14C-Tetraethylammonium (TEA) by test compounds was studied in wild-type an... | US Patent US9663528 (2017) BindingDB Entry DOI: 10.7270/Q24F1SVF | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| POU domain, class 2, transcription factor 2 (Homo sapiens (Human)) | BDBM330060 ((1R,3S,4R,12aR)-7-hydroxy-3-methyl-6,8-dioxo-N-(2,...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 1.51E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Gilead Sciences, Inc. US Patent | Assay Description The dose dependent inhibition of OCT2 mediated uptake of a model substrate 14C-Tetraethylammonium (TEA) by test compounds was studied in wild-type an... | US Patent US10689399 (2020) | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Solute carrier family 22 member 2 (Homo sapiens (Human)) | BDBM330060 ((1R,3S,4R,12aR)-7-hydroxy-3-methyl-6,8-dioxo-N-(2,...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 1.51E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Gilead Sciences, Inc. US Patent | Assay Description Test compounds were serially diluted in DMSO and then spiked (2 μL) into in 0.4 mL KHB buffer containing wild-type or OCT2-transfected cells and... | US Patent US9732092 (2017) BindingDB Entry DOI: 10.7270/Q2SN0C2R | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||