

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM359512 4-(1-(2-chloro-6-(1- cyanocyclopropyl) benzoyl)-6-(3- methoxyazetidine-1- carbonyl)-4,5,6,7- tetrahydro-1H- indazol-3-yl)-3- fluorobenzoic acid::US10221142, Example 13A-A::US10221142, Example 13A-B
SMILES: COC1CN(C1)C(=O)C1CCc2c(C1)n(nc2-c1ccc(cc1F)C(O)=O)C(=O)c1c(Cl)cccc1C1(CC1)C#N
InChI Key: InChIKey=YXVWEEAIKPTUMW-UHFFFAOYSA-N
Data: 2 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Nuclear receptor ROR-gamma (RORC) (Homo sapiens (Human)) | BDBM359512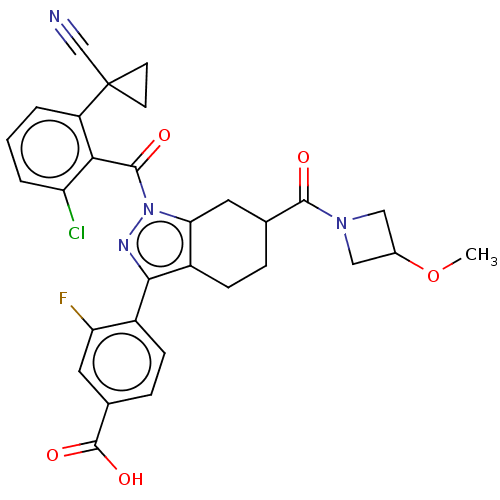 (4-(1-(2-chloro-6-(1- cyanocyclopropyl) benzoyl)-6-...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 3.60 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Corp. US Patent | Assay Description The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I... | US Patent US10221142 (2019) BindingDB Entry DOI: 10.7270/Q2M90BZ0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nuclear receptor ROR-gamma (RORC) (Homo sapiens (Human)) | BDBM359512 (4-(1-(2-chloro-6-(1- cyanocyclopropyl) benzoyl)-6-...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 642 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck Sharp & Dohme Corp. US Patent | Assay Description The compounds of the invention inhibit RORgammaT activity. Activation of RORgammaT activity can be measured using, e.g., biochemical TR-FRET assay. I... | US Patent US10221142 (2019) BindingDB Entry DOI: 10.7270/Q2M90BZ0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||