

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
null
SMILES: COc1ccc(CS(=O)(=O)[C@@H]2C[C@H](N(C2)C(=O)[C@@H](Cc2cccc(Cl)c2)Oc2cc(Cl)cc(Cl)c2)C(=O)N[C@@H](C(C)C)C(=O)C(F)(F)C(=O)NCC(F)(F)F)cc1
InChI Key: InChIKey=OBSDQVAMXNUIKC-ITGKQZKFSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Serine protease HTRA1 (Homo sapiens (Human)) | BDBM496988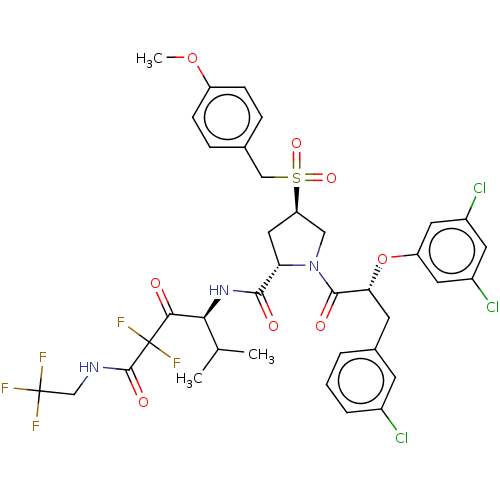 (US11001555, Example 11) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | PC cid PC sid UniChem | US Patent | n/a | n/a | 0.694 | n/a | n/a | n/a | n/a | n/a | n/a |
Hoffman-La Roche Inc. US Patent | Assay Description Enzyme activity is measured by observing the increase in fluorescence intensity caused by cleavage of a peptide substrate containing a fluorophore, w... | US Patent US11001555 (2021) | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||