

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
null
SMILES: NCCCC[C@@H](NC(=O)c1cccc2c3C[C@@]4(O)C5Cc6ccc(O)c7O[C@@H](c3[nH]c12)C4(CCN5CC1CC1)c67)C(O)=O
InChI Key: InChIKey=XVHSCIPOAOUAJI-RLDRNQAHSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50036136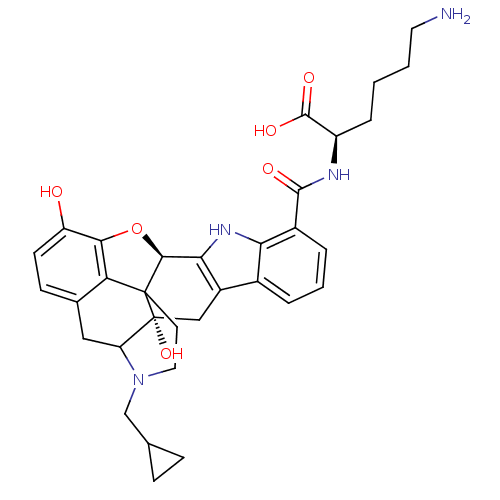 (6-amino-2-[22-cyclopropylmethyl-2,16-dihydroxy-(2S...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Minnesota Curated by ChEMBL | Assay Description The opioid receptor affinity(Ki) was evaluated by competition with [3H]-DPDPE (delta 1) on guinea pig brain membranes | J Med Chem 38: 402-7 (1995) BindingDB Entry DOI: 10.7270/Q22N52W5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Delta-type opioid receptor (Homo sapiens (Human)) | BDBM50036136 (6-amino-2-[22-cyclopropylmethyl-2,16-dihydroxy-(2S...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 0.75 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Minnesota Curated by ChEMBL | Assay Description The opioid receptor affinity(Ki) was evaluated by competition with [3H]DSLET (delta 2) on guinea pig brain membranes | J Med Chem 38: 402-7 (1995) BindingDB Entry DOI: 10.7270/Q22N52W5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Kappa-type opioid receptor (Cavia porcellus (domestic guinea pig)) | BDBM50036136 (6-amino-2-[22-cyclopropylmethyl-2,16-dihydroxy-(2S...) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 41 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Minnesota Curated by ChEMBL | Assay Description The opioid receptor affinity(Ki) was evaluated by competition with 9 (Opioid receptor kappa 1) on guinea pig brain membranes | J Med Chem 38: 402-7 (1995) BindingDB Entry DOI: 10.7270/Q22N52W5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Mu-type opioid receptor (GUINEA PIG) | BDBM50036136 (6-amino-2-[22-cyclopropylmethyl-2,16-dihydroxy-(2S...) | UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | 219 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Minnesota Curated by ChEMBL | Assay Description The opioid receptor affinity(Ki) was evaluated by competition with [3H]DAMGO (mu) on guinea pig brain membranes | J Med Chem 38: 402-7 (1995) BindingDB Entry DOI: 10.7270/Q22N52W5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||