

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50044676 CHEMBL439044
SMILES: [H][C@]1(NC(=O)[C@H](Cc2ccc(O)cc2)NC(=O)CCSSC[C@H](NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCC(N)=O)NC1=O)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CC(C)C)C(=O)NCC(N)=O)[C@@H](C)CC
InChI Key: InChIKey=GTYWGUNQAMYZPF-QPLNMOKZSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Oxytocin receptor (Homo sapiens (Human)) | BDBM50044676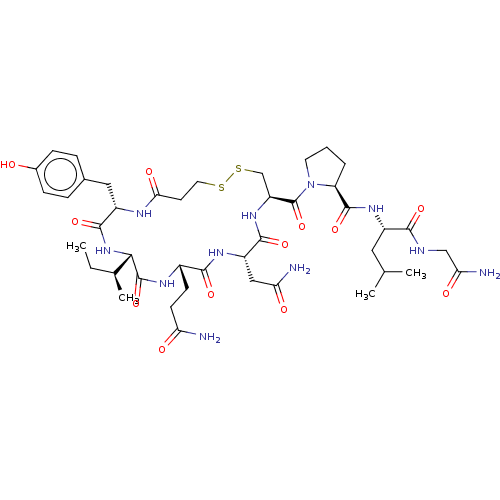 (CHEMBL439044) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.330 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Displacement of [3H]8-arginine-vasopressin from human oxytocin receptor expressed in CHO cell membrane incubated for 1 hr by liquid scintillation cou... | Bioorg Med Chem 24: 3513-20 (2016) BindingDB Entry DOI: 10.7270/Q2WH2RXT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxytocin receptor (Homo sapiens (Human)) | BDBM50044676 (CHEMBL439044) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a |
Ferring Research Institute Inc. Curated by ChEMBL | Assay Description Agonist activity at human oxytocin receptor expressed in CHO-K1 cells after 5 hrs by firefly luciferase reporter gene assay | J Med Chem 57: 5306-17 (2014) Article DOI: 10.1021/jm500365s BindingDB Entry DOI: 10.7270/Q2WD426V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V1a receptor (Homo sapiens (Human)) | BDBM50044676 (CHEMBL439044) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 21 | n/a | n/a | n/a | n/a |
Ferring Research Institute Inc. Curated by ChEMBL | Assay Description Agonist activity at human vasopressin V1a expressed in HEK293 cells after 5 hrs by firefly luciferase reporter gene assay | J Med Chem 57: 5306-17 (2014) Article DOI: 10.1021/jm500365s BindingDB Entry DOI: 10.7270/Q2WD426V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V1a receptor (Homo sapiens (Human)) | BDBM50044676 (CHEMBL439044) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Ferring Research Institute Inc. Curated by ChEMBL | Assay Description Antagonist activity at human vasopressin V1a expressed in AVP-stimulated HEK293 cells after 5 hrs by firefly luciferase reporter gene assay | J Med Chem 57: 5306-17 (2014) Article DOI: 10.1021/jm500365s BindingDB Entry DOI: 10.7270/Q2WD426V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V1b receptor (Homo sapiens (Human)) | BDBM50044676 (CHEMBL439044) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 180 | n/a | n/a | n/a | n/a |
Ferring Research Institute Inc. Curated by ChEMBL | Assay Description Agonist activity at human vasopressin V1b expressed in HEK293 cells after 5 hrs by firefly luciferase reporter gene assay | J Med Chem 57: 5306-17 (2014) Article DOI: 10.1021/jm500365s BindingDB Entry DOI: 10.7270/Q2WD426V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V1a receptor (Homo sapiens (Human)) | BDBM50044676 (CHEMBL439044) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article | n/a | n/a | n/a | n/a | 41 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at vasopressin 1a receptor (unknown origin) | Citation and Details Article DOI: 10.1016/j.bmcl.2017.12.027 BindingDB Entry DOI: 10.7270/Q2VT1VPB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V2 receptor (Homo sapiens (Human)) | BDBM50044676 (CHEMBL439044) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 3.5 | n/a | n/a | n/a | n/a |
Ferring Research Institute Inc. Curated by ChEMBL | Assay Description Agonist activity at human vasopressin V2 expressed in HEK293 cells after 5 hrs by firefly luciferase reporter gene assay | J Med Chem 57: 5306-17 (2014) Article DOI: 10.1021/jm500365s BindingDB Entry DOI: 10.7270/Q2WD426V | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V1a receptor (Homo sapiens (Human)) | BDBM50044676 (CHEMBL439044) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.10E+4 | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd. Curated by ChEMBL | Assay Description Agonist activity at human V1a receptor expressed in CHO cells assessed as calcium mobilization measured after 30 mins by Fluo-4-AM dye based FLIPR as... | Bioorg Med Chem Lett 27: 2331-2335 (2017) Article DOI: 10.1016/j.bmcl.2017.04.030 BindingDB Entry DOI: 10.7270/Q2BR8V9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Vasopressin V1a receptor (Homo sapiens (Human)) | BDBM50044676 (CHEMBL439044) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 3.20E+3 | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd. Curated by ChEMBL | Assay Description Agonist activity at human V1b receptor expressed in CHO cells assessed as calcium mobilization measured after 30 mins by Fluo-4-AM dye based FLIPR as... | Bioorg Med Chem Lett 27: 2331-2335 (2017) Article DOI: 10.1016/j.bmcl.2017.04.030 BindingDB Entry DOI: 10.7270/Q2BR8V9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxytocin receptor (Homo sapiens (Human)) | BDBM50044676 (CHEMBL439044) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | n/a | n/a | 9.10 | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company, Ltd. Curated by ChEMBL | Assay Description Agonist activity at human OTR expressed in CHO cells assessed as calcium mobilization measured after 30 mins by Fluo-4-AM dye based FLIPR assay | Bioorg Med Chem Lett 27: 2331-2335 (2017) Article DOI: 10.1016/j.bmcl.2017.04.030 BindingDB Entry DOI: 10.7270/Q2BR8V9Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxytocin receptor (Homo sapiens (Human)) | BDBM50044676 (CHEMBL439044) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article | n/a | n/a | n/a | n/a | 0.0400 | n/a | n/a | n/a | n/a |
TBA | Assay Description Agonist activity at human OTR expressed in CHO cells assessed as increase in calcium flux after 60 to 120 mins by fluo-4 dye based FLIPR assay | Citation and Details Article DOI: 10.1016/j.bmcl.2017.12.027 BindingDB Entry DOI: 10.7270/Q2VT1VPB | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Oxytocin receptor (Homo sapiens (Human)) | BDBM50044676 (CHEMBL439044) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | n/a | n/a | 0.0400 | n/a | n/a | n/a | n/a |
Pfizer Inc. Curated by ChEMBL | Assay Description Agonist activity at human oxytocin receptor expressed in CHO cells assessed as increase in intracellular calcium flux measured for 90 sec by fluo-4 d... | Bioorg Med Chem 24: 3513-20 (2016) BindingDB Entry DOI: 10.7270/Q2WH2RXT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||