

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50049385 5-Hydroxy-2-phenyl-chromen-4-one::5-Hydroxyflavone::5-hydroxy-2-phenyl-4H-chromen-4-one::5-hydroxy-flavone::CHEMBL16807::NSC-26745
SMILES: Oc1cccc2oc(cc(=O)c12)-c1ccccc1
InChI Key: InChIKey=IYBLVRRCNVHZQJ-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50049385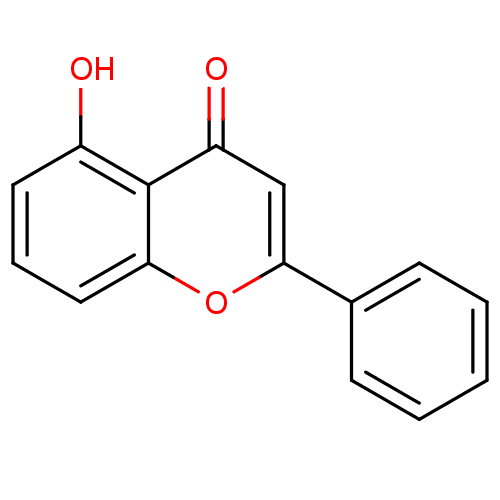 (5-Hydroxy-2-phenyl-chromen-4-one | 5-Hydroxyflavon...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2.17E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Displacement of specific [3H]-PIA binding from adenosine A1 receptor in rat brain membranes. | J Med Chem 39: 781-8 (1996) Article DOI: 10.1021/jm950661k BindingDB Entry DOI: 10.7270/Q2M32TV4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine receptor A1 (Rattus norvegicus (rat)) | BDBM50049385 (5-Hydroxy-2-phenyl-chromen-4-one | 5-Hydroxyflavon...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 2.19E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Ability to displace [3H]N6-phenylisopropyladenosine binding from adenosine A1 receptor. | J Med Chem 41: 46-52 (1998) Checked by Author Article DOI: 10.1021/jm970446z BindingDB Entry DOI: 10.7270/Q2NC62QT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine Receptors A2a (A2a) (Rattus norvegicus (rat)) | BDBM50049385 (5-Hydroxy-2-phenyl-chromen-4-one | 5-Hydroxyflavon...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 6.17E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Ability to displace [3H]-CGS- 21680 binding from adenosine A2A receptor. | J Med Chem 41: 46-52 (1998) Checked by Author Article DOI: 10.1021/jm970446z BindingDB Entry DOI: 10.7270/Q2NC62QT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Adenosine Receptors A2a (A2a) (Rattus norvegicus (rat)) | BDBM50049385 (5-Hydroxy-2-phenyl-chromen-4-one | 5-Hydroxyflavon...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 6.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
National Institute of Diabetes Curated by ChEMBL | Assay Description Affinity at Adenosine A2A receptor in rat striatal membranes by [3H]- CGS 21680 displacement. | J Med Chem 39: 781-8 (1996) Article DOI: 10.1021/jm950661k BindingDB Entry DOI: 10.7270/Q2M32TV4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 1A (Homo sapiens (Human)) | BDBM50049385 (5-Hydroxy-2-phenyl-chromen-4-one | 5-Hydroxyflavon...) | PDB MMDB Reactome pathway KEGG B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 310 | n/a | n/a | n/a | n/a | n/a | n/a |
Xavier University of Louisiana Curated by ChEMBL | Assay Description Inhibition of CYP1A2 (unknown origin)-mediated demethylation of resorufin methyl ether after 5 mins by spectrofluorimetric analysis | J Med Chem 56: 4082-92 (2013) Article DOI: 10.1021/jm4003654 BindingDB Entry DOI: 10.7270/Q2CN758W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 1A1 (Homo sapiens (Human)) | BDBM50049385 (5-Hydroxy-2-phenyl-chromen-4-one | 5-Hydroxyflavon...) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 4.03E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Xavier University of Louisiana Curated by ChEMBL | Assay Description Inhibition of CYP1A1 (unknown origin)-mediated deethylation of resorufin ethyl ether after 5 mins by spectrofluorimetric analysis | J Med Chem 56: 4082-92 (2013) Article DOI: 10.1021/jm4003654 BindingDB Entry DOI: 10.7270/Q2CN758W | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen Receptor (Homo sapiens (Human)) | BDBM50049385 (5-Hydroxy-2-phenyl-chromen-4-one | 5-Hydroxyflavon...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 840 | n/a | n/a | n/a | n/a | n/a | n/a |
Meijo University Curated by ChEMBL | Assay Description Antagonist activity at human androgen receptor LBD expressed in human MDA-kb2 cells by luciferase reporter gene assay | Bioorg Med Chem 21: 2968-74 (2013) Article DOI: 10.1016/j.bmc.2013.03.060 BindingDB Entry DOI: 10.7270/Q2FT8NFN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen Receptor (Homo sapiens (Human)) | BDBM50049385 (5-Hydroxy-2-phenyl-chromen-4-one | 5-Hydroxyflavon...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 832 | n/a | n/a | n/a | n/a | n/a | n/a |
Meijo University Curated by ChEMBL | Assay Description Antagonist activity at human androgen receptor LBD expressed in human MDA-kb2 cells by luciferase reporter gene assay | Bioorg Med Chem 21: 2968-74 (2013) Article DOI: 10.1016/j.bmc.2013.03.060 BindingDB Entry DOI: 10.7270/Q2FT8NFN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cytochrome P450 1B1 (Homo sapiens (Human)) | BDBM50049385 (5-Hydroxy-2-phenyl-chromen-4-one | 5-Hydroxyflavon...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 210 | n/a | n/a | n/a | n/a | n/a | n/a |
Laboratory of Medicinal Chemistry, Endocrinology and Nephrology Unit, CHU de Qu�bec - Research Center, Qu�bec, Qu�bec, Canada; Department of Molecular Medicine, Faculty of Medicine, Universit� Laval, Curated by ChEMBL | Assay Description Inhibition of human CYP1B1 expressed in Escherichia coli DH5alpha coexpressing human NADPH P450 reductase using 7-ethoxyresorufin as substrate in pre... | Eur J Med Chem 135: 296-306 (2017) Article DOI: 10.1016/j.ejmech.2017.04.042 BindingDB Entry DOI: 10.7270/Q26Q20QW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Androgen Receptor (Homo sapiens (Human)) | BDBM50049385 (5-Hydroxy-2-phenyl-chromen-4-one | 5-Hydroxyflavon...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 300 | n/a | n/a | n/a | n/a | n/a | n/a |
Meijo University Curated by ChEMBL | Assay Description Antagonist activity at androgen receptor in human MDA-kb2 cells assessed as inhibition of DHT-induced luciferase activity by luciferase reporter gene... | Bioorg Med Chem Lett 19: 4706-10 (2009) Article DOI: 10.1016/j.bmcl.2009.06.073 BindingDB Entry DOI: 10.7270/Q26T0MNQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Sialidase 2 (Homo sapiens (Human)) | BDBM50049385 (5-Hydroxy-2-phenyl-chromen-4-one | 5-Hydroxyflavon...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+6 | n/a | n/a | n/a | n/a | n/a | n/a |
Hokkaido University Curated by ChEMBL | Assay Description Inhibition of human Neu2 assessed as MuNANA substrate hydrolysis in presence of 0.1% Triton X-100 by discontinuous fluorimetric assay | Bioorg Med Chem 18: 1633-40 (2010) Article DOI: 10.1016/j.bmc.2009.12.062 BindingDB Entry DOI: 10.7270/Q26110F1 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||