

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50064653 2-Chloro-benzenesulfonic acid 3-methyl-5-[2-(methyl-pyridin-4-yl-amino)-ethoxy]-phenyl ester::CHEMBL289951
SMILES: CN(CCOc1cc(C)cc(OS(=O)(=O)c2ccccc2Cl)c1)c1ccncc1
InChI Key: InChIKey=MOAKYEVSWZWMBB-UHFFFAOYSA-N
Data: 7 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Prothrombin (Homo sapiens (Human)) | BDBM50064653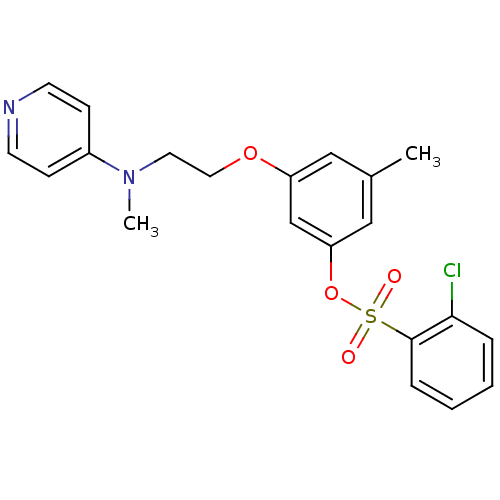 (2-Chloro-benzenesulfonic acid 3-methyl-5-[2-(methy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | PubMed | 11 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
3-Dimensional Pharmaceuticals Curated by ChEMBL | Assay Description Compound was tested in vitro for its ability to inhibit serine protease thrombin using succinyl-Ala-p-nitroanilide as substrate | Bioorg Med Chem Lett 8: 1595-600 (1999) BindingDB Entry DOI: 10.7270/Q20864GK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50064653 (2-Chloro-benzenesulfonic acid 3-methyl-5-[2-(methy...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | Article PubMed | 24 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
3-Dimensional Pharmaceuticals Inc. Curated by ChEMBL | Assay Description Compound was evaluated for the inhibition of thrombin | J Med Chem 41: 2068-75 (1998) Article DOI: 10.1021/jm970796l BindingDB Entry DOI: 10.7270/Q269748G | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Trypsin (Homo sapiens (Human)) | BDBM50064653 (2-Chloro-benzenesulfonic acid 3-methyl-5-[2-(methy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | PubMed | 1.10E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
3-Dimensional Pharmaceuticals Curated by ChEMBL | Assay Description Compound was tested in vitro for its ability to inhibit serine protease trypsin using benzoyl-Phe-Val-Arg-p-nitroanilide as substrate | Bioorg Med Chem Lett 8: 1595-600 (1999) BindingDB Entry DOI: 10.7270/Q20864GK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Chymotrypsin (Homo sapiens (Human)) | BDBM50064653 (2-Chloro-benzenesulfonic acid 3-methyl-5-[2-(methy...) | PDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | PubMed | 4.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
3-Dimensional Pharmaceuticals Curated by ChEMBL | Assay Description Compound was tested in vitro for its ability to inhibit serine protease chymotrypsin using suc-Ala-Ala-Pro-Phe-p-nitroanilide as substrate | Bioorg Med Chem Lett 8: 1595-600 (1999) BindingDB Entry DOI: 10.7270/Q20864GK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Coagulation factor X (Homo sapiens (Human)) | BDBM50064653 (2-Chloro-benzenesulfonic acid 3-methyl-5-[2-(methy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | PubMed | 1.60E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
3-Dimensional Pharmaceuticals Curated by ChEMBL | Assay Description Compound was tested in vitro for its ability to inhibit serine protease factor Xa using benzoyl-Ile-Glu-Gly-Arg-p-nitroanilide as substrate | Bioorg Med Chem Lett 8: 1595-600 (1999) BindingDB Entry DOI: 10.7270/Q20864GK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Urokinase-type plasminogen activator (Homo sapiens (Human)) | BDBM50064653 (2-Chloro-benzenesulfonic acid 3-methyl-5-[2-(methy...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | PubMed | >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
3-Dimensional Pharmaceuticals Curated by ChEMBL | Assay Description Compound was tested in vitro for its ability to inhibit serine protease urokinase using carbobenzyloxy-Phe-Val-Arg-pnitroanilide as substrate | Bioorg Med Chem Lett 8: 1595-600 (1999) BindingDB Entry DOI: 10.7270/Q20864GK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasminogen (Homo sapiens (Human)) | BDBM50064653 (2-Chloro-benzenesulfonic acid 3-methyl-5-[2-(methy...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents | PubMed | >2.00E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
3-Dimensional Pharmaceuticals Curated by ChEMBL | Assay Description Compound was tested in vitro for its ability to inhibit serine protease plasmin using tosyl-Gly-Pro-Lys-p-nitroanilide as substrate | Bioorg Med Chem Lett 8: 1595-600 (1999) BindingDB Entry DOI: 10.7270/Q20864GK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||