 Found 69 hits for monomerid = 50067678
Found 69 hits for monomerid = 50067678 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Sex hormone-binding globulin
(Homo sapiens (Human)) | BDBM50067678
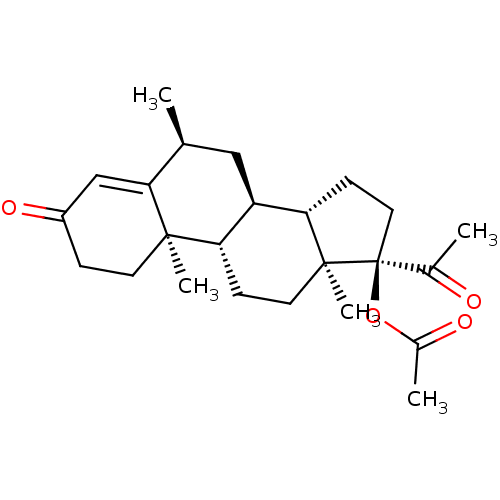
((6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-di...)Show SMILES C[C@H]1C[C@H]2[C@@H]3CC[C@](OC(C)=O)(C(C)=O)[C@@]3(C)CC[C@@H]2[C@@]2(C)CCC(=O)C=C12 |r,t:28| Show InChI InChI=1S/C24H34O4/c1-14-12-18-19(22(4)9-6-17(27)13-21(14)22)7-10-23(5)20(18)8-11-24(23,15(2)25)28-16(3)26/h13-14,18-20H,6-12H2,1-5H3/t14-,18+,19-,20-,22+,23-,24-/m0/s1 | PDB
MMDB
NCI pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 631 | n/a | n/a | n/a | n/a | n/a |
University of British Columbia
Curated by ChEMBL
| Assay Description
Displacement of [3H]5alpha dihydrotestosterone from human sex hormone binding globulin |
J Med Chem 51: 2047-56 (2008)
Article DOI: 10.1021/jm7011485
BindingDB Entry DOI: 10.7270/Q2RX9DC2 |
More data for this
Ligand-Target Pair | |
Progesterone receptor
(Homo sapiens (Human)) | BDBM50067678

((6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-di...)Show SMILES C[C@H]1C[C@H]2[C@@H]3CC[C@](OC(C)=O)(C(C)=O)[C@@]3(C)CC[C@@H]2[C@@]2(C)CCC(=O)C=C12 |r,t:28| Show InChI InChI=1S/C24H34O4/c1-14-12-18-19(22(4)9-6-17(27)13-21(14)22)7-10-23(5)20(18)8-11-24(23,15(2)25)28-16(3)26/h13-14,18-20H,6-12H2,1-5H3/t14-,18+,19-,20-,22+,23-,24-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
Article
PubMed
| n/a | n/a | n/a | n/a | 0.5 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Agonistic activity measures the ability to induce alkaline phosphatase in T47D human breast cancer cell line |
Bioorg Med Chem Lett 14: 2185-9 (2004)
Article DOI: 10.1016/j.bmcl.2004.02.054
BindingDB Entry DOI: 10.7270/Q2X34Z1K |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Mus musculus) | BDBM50067678

((6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-di...)Show SMILES C[C@H]1C[C@H]2[C@@H]3CC[C@](OC(C)=O)(C(C)=O)[C@@]3(C)CC[C@@H]2[C@@]2(C)CCC(=O)C=C12 |r,t:28| Show InChI InChI=1S/C24H34O4/c1-14-12-18-19(22(4)9-6-17(27)13-21(14)22)7-10-23(5)20(18)8-11-24(23,15(2)25)28-16(3)26/h13-14,18-20H,6-12H2,1-5H3/t14-,18+,19-,20-,22+,23-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | 6.10 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Effective concentration against Androgen receptor |
Bioorg Med Chem Lett 13: 1313-6 (2003)
BindingDB Entry DOI: 10.7270/Q2SN09HR |
More data for this
Ligand-Target Pair | |
Glucocorticoid receptor
(Homo sapiens (Human)) | BDBM50067678

((6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-di...)Show SMILES C[C@H]1C[C@H]2[C@@H]3CC[C@](OC(C)=O)(C(C)=O)[C@@]3(C)CC[C@@H]2[C@@]2(C)CCC(=O)C=C12 |r,t:28| Show InChI InChI=1S/C24H34O4/c1-14-12-18-19(22(4)9-6-17(27)13-21(14)22)7-10-23(5)20(18)8-11-24(23,15(2)25)28-16(3)26/h13-14,18-20H,6-12H2,1-5H3/t14-,18+,19-,20-,22+,23-,24-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | 10 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Effective concentration against GR (glucocorticoid receptor) |
Bioorg Med Chem Lett 13: 1313-6 (2003)
BindingDB Entry DOI: 10.7270/Q2SN09HR |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Rattus norvegicus (Rat)) | BDBM50067678

((6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-di...)Show SMILES C[C@H]1C[C@H]2[C@@H]3CC[C@](OC(C)=O)(C(C)=O)[C@@]3(C)CC[C@@H]2[C@@]2(C)CCC(=O)C=C12 |r,t:28| Show InChI InChI=1S/C24H34O4/c1-14-12-18-19(22(4)9-6-17(27)13-21(14)22)7-10-23(5)20(18)8-11-24(23,15(2)25)28-16(3)26/h13-14,18-20H,6-12H2,1-5H3/t14-,18+,19-,20-,22+,23-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 794 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Basel
Curated by ChEMBL
| Assay Description
Inhibitory concentration against recombinant rat androgen receptor expressed in Escherichia coli using [3H]methyltrienolone (R 1881) |
J Med Chem 48: 5666-74 (2005)
Article DOI: 10.1021/jm050403f
BindingDB Entry DOI: 10.7270/Q2TM7CBZ |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Rattus norvegicus (Rat)) | BDBM50067678

((6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-di...)Show SMILES C[C@H]1C[C@H]2[C@@H]3CC[C@](OC(C)=O)(C(C)=O)[C@@]3(C)CC[C@@H]2[C@@]2(C)CCC(=O)C=C12 |r,t:28| Show InChI InChI=1S/C24H34O4/c1-14-12-18-19(22(4)9-6-17(27)13-21(14)22)7-10-23(5)20(18)8-11-24(23,15(2)25)28-16(3)26/h13-14,18-20H,6-12H2,1-5H3/t14-,18+,19-,20-,22+,23-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 794 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Basel
Curated by ChEMBL
| Assay Description
Inhibitory concentration against recombinant rat androgen receptor expressed in Escherichia coli using [3H]methyltrienolone (R 1881) |
J Med Chem 48: 5666-74 (2005)
Article DOI: 10.1021/jm050403f
BindingDB Entry DOI: 10.7270/Q2TM7CBZ |
More data for this
Ligand-Target Pair | |
Progesterone receptor
(Homo sapiens (Human)) | BDBM50067678

((6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-di...)Show SMILES C[C@H]1C[C@H]2[C@@H]3CC[C@](OC(C)=O)(C(C)=O)[C@@]3(C)CC[C@@H]2[C@@]2(C)CCC(=O)C=C12 |r,t:28| Show InChI InChI=1S/C24H34O4/c1-14-12-18-19(22(4)9-6-17(27)13-21(14)22)7-10-23(5)20(18)8-11-24(23,15(2)25)28-16(3)26/h13-14,18-20H,6-12H2,1-5H3/t14-,18+,19-,20-,22+,23-,24-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
PubMed
| n/a | n/a | n/a | n/a | 0.100 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Effective concentration on alkaline phosphatase activity in human T47D breast carcinoma cell line. |
Bioorg Med Chem Lett 13: 1313-6 (2003)
BindingDB Entry DOI: 10.7270/Q2SN09HR |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Mus musculus) | BDBM50067678

((6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-di...)Show SMILES C[C@H]1C[C@H]2[C@@H]3CC[C@](OC(C)=O)(C(C)=O)[C@@]3(C)CC[C@@H]2[C@@]2(C)CCC(=O)C=C12 |r,t:28| Show InChI InChI=1S/C24H34O4/c1-14-12-18-19(22(4)9-6-17(27)13-21(14)22)7-10-23(5)20(18)8-11-24(23,15(2)25)28-16(3)26/h13-14,18-20H,6-12H2,1-5H3/t14-,18+,19-,20-,22+,23-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | 6.10 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Effective concentration on alkaline phosphatase activity in human T47D breast carcinoma cell line. |
Bioorg Med Chem Lett 13: 1313-6 (2003)
BindingDB Entry DOI: 10.7270/Q2SN09HR |
More data for this
Ligand-Target Pair | |
Glucocorticoid receptor
(Homo sapiens (Human)) | BDBM50067678

((6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-di...)Show SMILES C[C@H]1C[C@H]2[C@@H]3CC[C@](OC(C)=O)(C(C)=O)[C@@]3(C)CC[C@@H]2[C@@]2(C)CCC(=O)C=C12 |r,t:28| Show InChI InChI=1S/C24H34O4/c1-14-12-18-19(22(4)9-6-17(27)13-21(14)22)7-10-23(5)20(18)8-11-24(23,15(2)25)28-16(3)26/h13-14,18-20H,6-12H2,1-5H3/t14-,18+,19-,20-,22+,23-,24-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | n/a | n/a | 10 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Effective concentration against Androgen receptor |
Bioorg Med Chem Lett 13: 1313-6 (2003)
BindingDB Entry DOI: 10.7270/Q2SN09HR |
More data for this
Ligand-Target Pair | |
Progesterone receptor
(Homo sapiens (Human)) | BDBM50067678

((6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-di...)Show SMILES C[C@H]1C[C@H]2[C@@H]3CC[C@](OC(C)=O)(C(C)=O)[C@@]3(C)CC[C@@H]2[C@@]2(C)CCC(=O)C=C12 |r,t:28| Show InChI InChI=1S/C24H34O4/c1-14-12-18-19(22(4)9-6-17(27)13-21(14)22)7-10-23(5)20(18)8-11-24(23,15(2)25)28-16(3)26/h13-14,18-20H,6-12H2,1-5H3/t14-,18+,19-,20-,22+,23-,24-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
PubMed
| n/a | n/a | n/a | n/a | 0.120 | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Effective concentration against PR (progesterone receptor) |
Bioorg Med Chem Lett 13: 1313-6 (2003)
BindingDB Entry DOI: 10.7270/Q2SN09HR |
More data for this
Ligand-Target Pair | |
Progesterone receptor
(Homo sapiens (Human)) | BDBM50067678

((6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-di...)Show SMILES C[C@H]1C[C@H]2[C@@H]3CC[C@](OC(C)=O)(C(C)=O)[C@@]3(C)CC[C@@H]2[C@@]2(C)CCC(=O)C=C12 |r,t:28| Show InChI InChI=1S/C24H34O4/c1-14-12-18-19(22(4)9-6-17(27)13-21(14)22)7-10-23(5)20(18)8-11-24(23,15(2)25)28-16(3)26/h13-14,18-20H,6-12H2,1-5H3/t14-,18+,19-,20-,22+,23-,24-/m0/s1 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| DrugBank
PubMed
| n/a | n/a | 11 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibition of 3 nM [3H]R5020 binding to progesterone receptor in human T47D cells |
Bioorg Med Chem Lett 13: 1317-20 (2003)
BindingDB Entry DOI: 10.7270/Q2WS8SNP |
More data for this
Ligand-Target Pair | |
Progesterone receptor
(Rattus norvegicus) | BDBM50067678

((6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-di...)Show SMILES C[C@H]1C[C@H]2[C@@H]3CC[C@](OC(C)=O)(C(C)=O)[C@@]3(C)CC[C@@H]2[C@@]2(C)CCC(=O)C=C12 |r,t:28| Show InChI InChI=1S/C24H34O4/c1-14-12-18-19(22(4)9-6-17(27)13-21(14)22)7-10-23(5)20(18)8-11-24(23,15(2)25)28-16(3)26/h13-14,18-20H,6-12H2,1-5H3/t14-,18+,19-,20-,22+,23-,24-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| PubMed
| n/a | n/a | 67.3 | n/a | n/a | n/a | n/a | n/a | n/a |
Wyeth Research
Curated by ChEMBL
| Assay Description
Inhibitory progestational activity on oral administration in uterine C3 model |
Bioorg Med Chem Lett 13: 1317-20 (2003)
BindingDB Entry DOI: 10.7270/Q2WS8SNP |
More data for this
Ligand-Target Pair | |
Bile salt export pump
(Homo sapiens (Human)) | BDBM50067678

((6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-di...)Show SMILES C[C@H]1C[C@H]2[C@@H]3CC[C@](OC(C)=O)(C(C)=O)[C@@]3(C)CC[C@@H]2[C@@]2(C)CCC(=O)C=C12 |r,t:28| Show InChI InChI=1S/C24H34O4/c1-14-12-18-19(22(4)9-6-17(27)13-21(14)22)7-10-23(5)20(18)8-11-24(23,15(2)25)28-16(3)26/h13-14,18-20H,6-12H2,1-5H3/t14-,18+,19-,20-,22+,23-,24-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 1.57E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc
Curated by ChEMBL
| Assay Description
Inhibition of human BSEP overexpressed in Sf9 cell membrane vesicles assessed as uptake of [3H]-taurocholate in presence of ATP measured after 15 to ... |
Toxicol Sci 136: 216-41 (2013)
Article DOI: 10.1093/toxsci/kft176
BindingDB Entry DOI: 10.7270/Q2JM2D2D |
More data for this
Ligand-Target Pair | |
ATP-binding cassette sub-family C member 4
(Homo sapiens (Human)) | BDBM50067678

((6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-di...)Show SMILES C[C@H]1C[C@H]2[C@@H]3CC[C@](OC(C)=O)(C(C)=O)[C@@]3(C)CC[C@@H]2[C@@]2(C)CCC(=O)C=C12 |r,t:28| Show InChI InChI=1S/C24H34O4/c1-14-12-18-19(22(4)9-6-17(27)13-21(14)22)7-10-23(5)20(18)8-11-24(23,15(2)25)28-16(3)26/h13-14,18-20H,6-12H2,1-5H3/t14-,18+,19-,20-,22+,23-,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.25E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc
Curated by ChEMBL
| Assay Description
Inhibition of human MRP4 overexpressed in Sf9 cell membrane vesicles assessed as uptake of [3H]-estradiol-17beta-D-glucuronide in presence of ATP and... |
Toxicol Sci 136: 216-41 (2013)
Article DOI: 10.1093/toxsci/kft176
BindingDB Entry DOI: 10.7270/Q2JM2D2D |
More data for this
Ligand-Target Pair | |
ATP-binding cassette sub-family C member 3
(Homo sapiens (Human)) | BDBM50067678

((6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-di...)Show SMILES C[C@H]1C[C@H]2[C@@H]3CC[C@](OC(C)=O)(C(C)=O)[C@@]3(C)CC[C@@H]2[C@@]2(C)CCC(=O)C=C12 |r,t:28| Show InChI InChI=1S/C24H34O4/c1-14-12-18-19(22(4)9-6-17(27)13-21(14)22)7-10-23(5)20(18)8-11-24(23,15(2)25)28-16(3)26/h13-14,18-20H,6-12H2,1-5H3/t14-,18+,19-,20-,22+,23-,24-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.33E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc
Curated by ChEMBL
| Assay Description
Inhibition of human MRP3 overexpressed in Sf9 insect cell membrane vesicles assessed as uptake of [3H]-estradiol-17beta-D-glucuronide in presence of ... |
Toxicol Sci 136: 216-41 (2013)
Article DOI: 10.1093/toxsci/kft176
BindingDB Entry DOI: 10.7270/Q2JM2D2D |
More data for this
Ligand-Target Pair | |
ATP-binding cassette sub-family C member 2
(Homo sapiens (Human)) | BDBM50067678

((6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-di...)Show SMILES C[C@H]1C[C@H]2[C@@H]3CC[C@](OC(C)=O)(C(C)=O)[C@@]3(C)CC[C@@H]2[C@@]2(C)CCC(=O)C=C12 |r,t:28| Show InChI InChI=1S/C24H34O4/c1-14-12-18-19(22(4)9-6-17(27)13-21(14)22)7-10-23(5)20(18)8-11-24(23,15(2)25)28-16(3)26/h13-14,18-20H,6-12H2,1-5H3/t14-,18+,19-,20-,22+,23-,24-/m0/s1 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | >1.33E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
Amgen Inc
Curated by ChEMBL
| Assay Description
Inhibition of human MRP2 overexpressed in Sf9 cell membrane vesicles assessed as uptake of [3H]-estradiol-17beta-D-glucuronide in presence of ATP and... |
Toxicol Sci 136: 216-41 (2013)
Article DOI: 10.1093/toxsci/kft176
BindingDB Entry DOI: 10.7270/Q2JM2D2D |
More data for this
Ligand-Target Pair | |
Aldo-keto reductase family 1 member C2
(Homo sapiens (Human)) | BDBM50067678

((6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-di...)Show SMILES C[C@H]1C[C@H]2[C@@H]3CC[C@](OC(C)=O)(C(C)=O)[C@@]3(C)CC[C@@H]2[C@@]2(C)CCC(=O)C=C12 |r,t:28| Show InChI InChI=1S/C24H34O4/c1-14-12-18-19(22(4)9-6-17(27)13-21(14)22)7-10-23(5)20(18)8-11-24(23,15(2)25)28-16(3)26/h13-14,18-20H,6-12H2,1-5H3/t14-,18+,19-,20-,22+,23-,24-/m0/s1 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 3.98E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Texas Tech University Health Sciences Center
Curated by ChEMBL
| Assay Description
Inhibition of recombinant AKR1C2 (unknown origin) assessed as reduction in S-tetralol catalyzed oxidation in presence of NADPH by fluorescence assay |
ACS Med Chem Lett 7: 774-9 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00163
BindingDB Entry DOI: 10.7270/Q2QJ7MS2 |
More data for this
Ligand-Target Pair | |
Aldo-keto reductase family 1 member C3
(Homo sapiens (Human)) | BDBM50067678

((6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-di...)Show SMILES C[C@H]1C[C@H]2[C@@H]3CC[C@](OC(C)=O)(C(C)=O)[C@@]3(C)CC[C@@H]2[C@@]2(C)CCC(=O)C=C12 |r,t:28| Show InChI InChI=1S/C24H34O4/c1-14-12-18-19(22(4)9-6-17(27)13-21(14)22)7-10-23(5)20(18)8-11-24(23,15(2)25)28-16(3)26/h13-14,18-20H,6-12H2,1-5H3/t14-,18+,19-,20-,22+,23-,24-/m0/s1 | PDB
MMDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | 2.51E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Texas Tech University Health Sciences Center
Curated by ChEMBL
| Assay Description
Inhibition of recombinant N-terminal His-tagged AKR1C3 (unknown origin) assessed as rate of change of pyridine nucleotide absorbance in presence of N... |
ACS Med Chem Lett 7: 774-9 (2016)
Article DOI: 10.1021/acsmedchemlett.6b00163
BindingDB Entry DOI: 10.7270/Q2QJ7MS2 |
More data for this
Ligand-Target Pair | |
Androgen receptor
(Homo sapiens (Human)) | BDBM50067678

((6alpha)-17-(Acetyloxy)-6-methylpreg-4-ene-3,20-di...)Show SMILES C[C@H]1C[C@H]2[C@@H]3CC[C@](OC(C)=O)(C(C)=O)[C@@]3(C)CC[C@@H]2[C@@]2(C)CCC(=O)C=C12 |r,t:28| Show InChI InChI=1S/C24H34O4/c1-14-12-18-19(22(4)9-6-17(27)13-21(14)22)7-10-23(5)20(18)8-11-24(23,15(2)25)28-16(3)26/h13-14,18-20H,6-12H2,1-5H3/t14-,18+,19-,20-,22+,23-,24-/m0/s1 | PDB
MMDB
NCI pathway
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| Purchase
DrugBank
MCE
KEGG
PC cid
PC sid
UniChem
Patents
Similars
| Article
PubMed
| n/a | n/a | n/a | 1 | n/a | n/a | n/a | n/a | n/a |
TBA
| |
Citation and Details
Article DOI: 10.1021/acs.jmedchem.2c00716
BindingDB Entry DOI: 10.7270/Q2J1076H |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data