

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50081125 CHEMBL3421961
SMILES: [#6]-c1cc(-[#8])cc(-[#6])c1\[#6]=[#6]-1/[#6](=O)-[#7](-[#6]-c2ccccc2)-[#6](=O)-[#7](-[#6]-c2ccccc2)-[#6]-1=O
InChI Key: InChIKey=LUGQBJDYUPNAQQ-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Histone acetyltransferase p300 (Homo sapiens (Human)) | BDBM50081125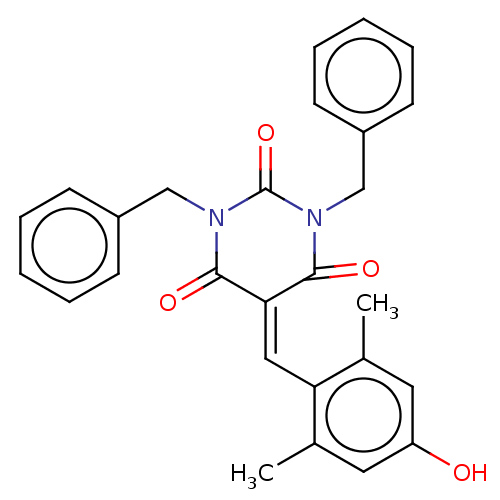 (CHEMBL3421961) | PDB MMDB UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 2.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Salerno Curated by ChEMBL | Assay Description Noncompetitive inhibition of P300 (unknown origin) using acetyl CoA as substrate after 15 mins by double reciprocal plot analysis | J Med Chem 58: 2779-98 (2015) Article DOI: 10.1021/jm5019687 BindingDB Entry DOI: 10.7270/Q2736SMP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone acetyltransferase p300 (Homo sapiens (Human)) | BDBM50081125 (CHEMBL3421961) | PDB MMDB UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | 9.40E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Salerno Curated by ChEMBL | Assay Description Noncompetitive inhibition of P300 (unknown origin) using biotinylated H3 as substrate after 15 mins by double reciprocal plot analysis | J Med Chem 58: 2779-98 (2015) Article DOI: 10.1021/jm5019687 BindingDB Entry DOI: 10.7270/Q2736SMP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Histone acetyltransferase p300 (Homo sapiens (Human)) | BDBM50081125 (CHEMBL3421961) | PDB MMDB UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 2.90E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Salerno Curated by ChEMBL | Assay Description Inhibition of recombinant P300 (unknown origin) | J Med Chem 58: 2779-98 (2015) Article DOI: 10.1021/jm5019687 BindingDB Entry DOI: 10.7270/Q2736SMP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| CREB-binding protein (Homo sapiens (Human)) | BDBM50081125 (CHEMBL3421961) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Universit£ degli Studi di Salerno Curated by ChEMBL | Assay Description Inhibition of recombinant CBP (unknown origin) | J Med Chem 58: 2779-98 (2015) Article DOI: 10.1021/jm5019687 BindingDB Entry DOI: 10.7270/Q2736SMP | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||