

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50093142 1,5-di{4-[4-amino(imino)methylaminobenzylcarbamoyl]hexahydro-1-pyrazinylcarbonyloxy}cyclooctane::1N-(3,3-diphenylpropyl)-4-[4-amino(imino)methylphenylcarboxamidomethyl]benzamide::APC-1390::CHEMBL46809::Derivative of piperazine-1-carboxylic acid 5-(piperazine-1-carbonyloxy)-cyclooctyl ester
SMILES: [#7]\[#6](-[#7])=[#7]/c1ccc(-[#6]-[#7]-[#6](=O)-[#7]-2-[#6]-[#6]-[#7](-[#6]-[#6]-2)-[#6](=O)-[#8]-[#6@H]-2-[#6]-[#6]-[#6]-[#6@H](-[#6]-[#6]-[#6]-2)-[#8]-[#6](=O)-[#7]-2-[#6]-[#6]-[#7](-[#6]-[#6]-2)-[#6](=O)-[#7]-[#6]-c2ccc(cc2)\[#7]=[#6](/[#7])-[#7])cc1
InChI Key: InChIKey=BYQFZIDMWZQLEL-RNPORBBMSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Tryptase (Homo sapiens (Human)) | BDBM50093142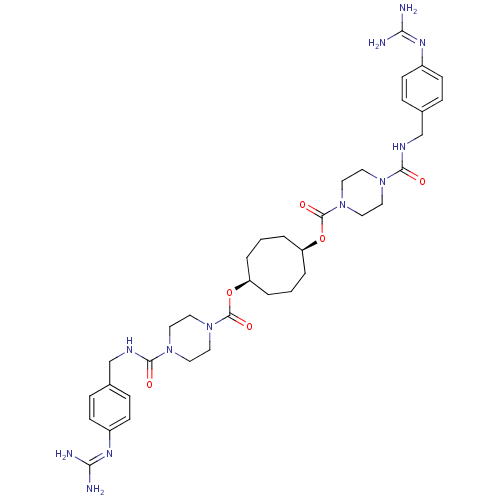 (1,5-di{4-[4-amino(imino)methylaminobenzylcarbamoyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 0.0700 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Inhibitory activity against human tryptase enzyme | Bioorg Med Chem Lett 11: 2325-30 (2001) BindingDB Entry DOI: 10.7270/Q21835S6 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Trypsin-1 (Homo sapiens (Human)) | BDBM50093142 (1,5-di{4-[4-amino(imino)methylaminobenzylcarbamoyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 3.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Inhibitory potency against trypsin | Bioorg Med Chem Lett 10: 2357-60 (2001) BindingDB Entry DOI: 10.7270/Q2NP23NX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Trypsin-1 (Homo sapiens (Human)) | BDBM50093142 (1,5-di{4-[4-amino(imino)methylaminobenzylcarbamoyl...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 3.90E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Compound was evaluated for its inhibitory potency against trypsin | Bioorg Med Chem Lett 10: 2361-6 (2001) BindingDB Entry DOI: 10.7270/Q2HX1BWR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50093142 (1,5-di{4-[4-amino(imino)methylaminobenzylcarbamoyl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 4.35E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Inhibitory potency against thrombin | Bioorg Med Chem Lett 10: 2357-60 (2001) BindingDB Entry DOI: 10.7270/Q2NP23NX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Prothrombin (Homo sapiens (Human)) | BDBM50093142 (1,5-di{4-[4-amino(imino)methylaminobenzylcarbamoyl...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 4.35E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Compound was evaluated for its inhibitory potency against thrombin | Bioorg Med Chem Lett 10: 2361-6 (2001) BindingDB Entry DOI: 10.7270/Q2HX1BWR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasminogen (Homo sapiens (Human)) | BDBM50093142 (1,5-di{4-[4-amino(imino)methylaminobenzylcarbamoyl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 4.94E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Compound was evaluated for its inhibitory potency against plasmin | Bioorg Med Chem Lett 10: 2361-6 (2001) BindingDB Entry DOI: 10.7270/Q2HX1BWR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Plasminogen (Homo sapiens (Human)) | BDBM50093142 (1,5-di{4-[4-amino(imino)methylaminobenzylcarbamoyl...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | 4.94E+5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Inhibitory potency against plasmin | Bioorg Med Chem Lett 10: 2357-60 (2001) BindingDB Entry DOI: 10.7270/Q2NP23NX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase (Homo sapiens (Human)) | BDBM50093142 (1,5-di{4-[4-amino(imino)methylaminobenzylcarbamoyl...) | KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | n/a | 0.0700 | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Dissociation cosntant of compound was evaluated from its IC50 against tryptase at different concentrations | Bioorg Med Chem Lett 10: 2357-60 (2001) BindingDB Entry DOI: 10.7270/Q2NP23NX | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Tryptase (Homo sapiens (Human)) | BDBM50093142 (1,5-di{4-[4-amino(imino)methylaminobenzylcarbamoyl...) | KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | PubMed | n/a | n/a | n/a | 0.0700 | n/a | n/a | n/a | n/a | n/a |
Axys Pharmaceuticals, Inc. Curated by ChEMBL | Assay Description Dissociation cosntant of compound was evaluated from its IC50 against tryptase at different concentrations | Bioorg Med Chem Lett 10: 2361-6 (2001) BindingDB Entry DOI: 10.7270/Q2HX1BWR | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||