

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50103962 (1R,3aS,5aR,5bR,7aR,9S,11aR,11bR,13aR,13bS)-1-Acetyl-9-hydroxy-5a,5b,8,8,11a-pentamethyl-icosahydro-cyclopenta[a]chrysene-3a-carboxylic acid::1-Acetyl-9-hydroxy-5alpha,5beta,8,8,11alpha-pentamethylicosahydro-3rHcyclopenta[alpha]chrysene-3alpha-carboxylic Acid::CHEMBL80460::platanic acid
SMILES: CC(=O)[C@@H]1CC[C@@]2(CC[C@]3(C)[C@H](CC[C@@H]4[C@@]5(C)CC[C@H](O)C(C)(C)[C@@H]5CC[C@@]34C)[C@@H]12)C(O)=O
InChI Key: InChIKey=RVMPLOSJMIQORE-FUAAEJBOSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50103962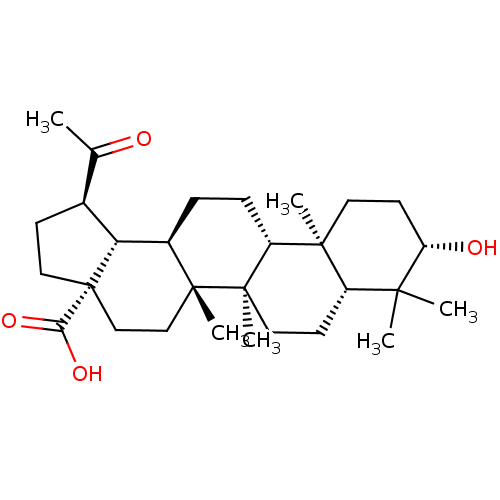 ((1R,3aS,5aR,5bR,7aR,9S,11aR,11bR,13aR,13bS)-1-Acet...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | 3.16E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Martin-Luther University Halle-Wittenberg Curated by ChEMBL | Assay Description Competitive inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 20 mins followed by substrate addition measu... | Eur J Med Chem 126: 652-668 (2017) BindingDB Entry DOI: 10.7270/Q2X0698K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50103962 ((1R,3aS,5aR,5bR,7aR,9S,11aR,11bR,13aR,13bS)-1-Acet...) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | 3.69E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Martin-Luther University Halle-Wittenberg Curated by ChEMBL | Assay Description Uncompetitive inhibition of electric eel AChE using acetylthiocholine iodide as substrate preincubated for 20 mins followed by substrate addition mea... | Eur J Med Chem 126: 652-668 (2017) BindingDB Entry DOI: 10.7270/Q2X0698K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cholinesterase (Equus caballus (Horse)) | BDBM50103962 ((1R,3aS,5aR,5bR,7aR,9S,11aR,11bR,13aR,13bS)-1-Acet...) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | PubMed | >5.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Martin-Luther University Halle-Wittenberg Curated by ChEMBL | Assay Description Competitive inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 20 mins followed by substrate addition meas... | Eur J Med Chem 126: 652-668 (2017) BindingDB Entry DOI: 10.7270/Q2X0698K | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protein kinase C, epsilon (Homo sapiens (Human)) | BDBM50103962 ((1R,3aS,5aR,5bR,7aR,9S,11aR,11bR,13aR,13bS)-1-Acet...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of PKC epsilon | J Nat Prod 57: 243-247 (1994) Article DOI: 10.1021/np50104a008 BindingDB Entry DOI: 10.7270/Q2TT4R0Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| PKC alpha and beta-2 (Homo sapiens (Human)) | BDBM50103962 ((1R,3aS,5aR,5bR,7aR,9S,11aR,11bR,13aR,13bS)-1-Acet...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | >1.50E+5 | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Inhibition of PKC beta2 | J Nat Prod 57: 243-247 (1994) Article DOI: 10.1021/np50104a008 BindingDB Entry DOI: 10.7270/Q2TT4R0Z | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| G-protein coupled bile acid receptor 1 (Homo sapiens (Human)) | BDBM50103962 ((1R,3aS,5aR,5bR,7aR,9S,11aR,11bR,13aR,13bS)-1-Acet...) | PDB Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 3.11E+3 | n/a | n/a | n/a | n/a |
Universite Louis Pasteur Curated by ChEMBL | Assay Description Agonist activity at TGR5 expressed in CHO cells by CRE-driven luciferase reporter gene assay | J Med Chem 53: 178-90 (2010) Article DOI: 10.1021/jm900872z BindingDB Entry DOI: 10.7270/Q2RJ4KDW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||