

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50119367 (3R)-N-[(2R)-3-(4-chlorophenyl)-1-{4-cyclohexyl-4-[(1-methyl-1H-1,2,3,4-tetrazol-5-yl)methyl]piperidin-1-yl}-1-oxopropan-2-yl]-1,2,3,4-tetrahydroisoquinoline-3-carboxamide::1,2,3,4-Tetrahydro-isoquinoline-3-carboxylic acid {1-(4-chloro-benzyl)-2-[4-cyclohexyl-4-(1-methyl-1H-tetrazol-5-ylmethyl)-piperidin-1-yl]-2-oxo-ethyl}-amide::CHEMBL140154
SMILES: Cn1nnnc1CC1(CCN(CC1)C(=O)[C@@H](Cc1ccc(Cl)cc1)NC(=O)[C@H]1Cc2ccccc2CN1)C1CCCCC1
InChI Key: InChIKey=IJYUZXAPJHBRJR-FQLXRVMXSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Melanocortin receptor 4 (Homo sapiens (Human)) | BDBM50119367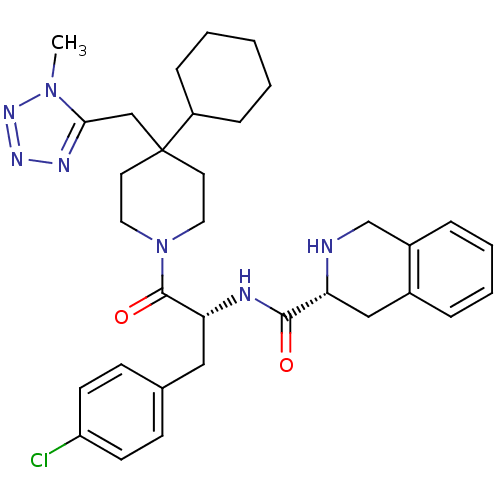 ((3R)-N-[(2R)-3-(4-chlorophenyl)-1-{4-cyclohexyl-4-...) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Merck & Co. Curated by ChEMBL | Assay Description Evaluated for binding affinity against Melanocortin-4 receptor by displacing [125I]-NDP-alpha-MSH radioligand expressed in CHO cells | J Med Chem 45: 4589-93 (2002) BindingDB Entry DOI: 10.7270/Q2GT5MH9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Melanocortin receptor 3 (Homo sapiens (Human)) | BDBM50119367 ((3R)-N-[(2R)-3-(4-chlorophenyl)-1-{4-cyclohexyl-4-...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 59 | n/a | n/a | n/a | n/a |
Merck & Co. Curated by ChEMBL | Assay Description Evaluated for Functional Activity at MC3R as effective concentration at 50% maximum CMP accumulation | J Med Chem 45: 4589-93 (2002) BindingDB Entry DOI: 10.7270/Q2GT5MH9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Melanocortin receptor 4 (Homo sapiens (Human)) | BDBM50119367 ((3R)-N-[(2R)-3-(4-chlorophenyl)-1-{4-cyclohexyl-4-...) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 0.300 | n/a | n/a | n/a | n/a | n/a | n/a |
Neurocrine Biosciences, Inc. Curated by ChEMBL | Assay Description Agonist activity at human MC4 receptor expressed in HEK293 cells assessed as stimulation of cAMP production | J Med Chem 47: 6821-30 (2004) Article DOI: 10.1021/jm049278i BindingDB Entry DOI: 10.7270/Q2610ZSQ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Melanocortin receptor 5 (MC5R) (Homo sapiens (Human)) | BDBM50119367 ((3R)-N-[(2R)-3-(4-chlorophenyl)-1-{4-cyclohexyl-4-...) | UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 127 | n/a | n/a | n/a | n/a |
Merck & Co. Curated by ChEMBL | Assay Description Evaluated for Functional Activity at MC5R as effective concentration at 50% maximum CMP accumulation | J Med Chem 45: 4589-93 (2002) BindingDB Entry DOI: 10.7270/Q2GT5MH9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Melanocortin receptor 4 (Homo sapiens (Human)) | BDBM50119367 ((3R)-N-[(2R)-3-(4-chlorophenyl)-1-{4-cyclohexyl-4-...) | PDB KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | PubMed | n/a | n/a | n/a | n/a | 0.600 | n/a | n/a | n/a | n/a |
Merck & Co. Curated by ChEMBL | Assay Description Evaluated for Functional Activity at Melanocortin-4 receptor as effective concentration at 50% maximum CMP accumulation | J Med Chem 45: 4589-93 (2002) BindingDB Entry DOI: 10.7270/Q2GT5MH9 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||