 Found 4 hits for monomerid = 50205313
Found 4 hits for monomerid = 50205313 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
Vasopressin V1b receptor
(RAT) | BDBM50205313
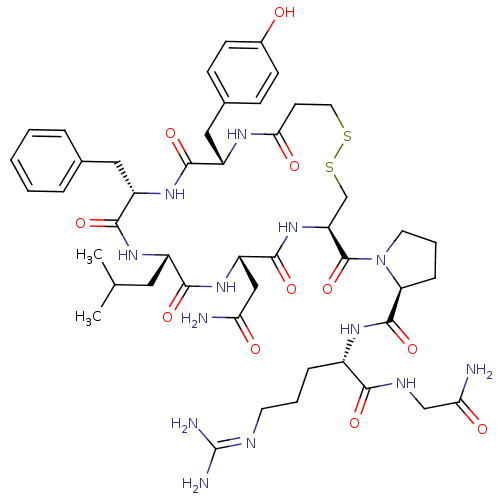
(CHEMBL265859 | d[Leu4]AVP)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@@H]-1-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccccc2)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6](=O)-[#6]-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C47H67N13O11S2/c1-26(2)20-31-41(66)58-34(23-37(48)62)44(69)59-35(46(71)60-18-7-11-36(60)45(70)55-30(10-6-17-52-47(50)51)40(65)53-24-38(49)63)25-73-72-19-16-39(64)54-32(22-28-12-14-29(61)15-13-28)42(67)57-33(43(68)56-31)21-27-8-4-3-5-9-27/h3-5,8-9,12-15,26,30-36,61H,6-7,10-11,16-25H2,1-2H3,(H2,48,62)(H2,49,63)(H,53,65)(H,54,64)(H,55,70)(H,56,68)(H,57,67)(H,58,66)(H,59,69)(H4,50,51,52)/t30-,31-,32-,33-,34-,35-,36-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 0.0400 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from rat vasopressin V1b receptor expressed in At-T20 cells |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V2 receptor
(Rattus norvegicus (Rat)) | BDBM50205313

(CHEMBL265859 | d[Leu4]AVP)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@@H]-1-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccccc2)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6](=O)-[#6]-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C47H67N13O11S2/c1-26(2)20-31-41(66)58-34(23-37(48)62)44(69)59-35(46(71)60-18-7-11-36(60)45(70)55-30(10-6-17-52-47(50)51)40(65)53-24-38(49)63)25-73-72-19-16-39(64)54-32(22-28-12-14-29(61)15-13-28)42(67)57-33(43(68)56-31)21-27-8-4-3-5-9-27/h3-5,8-9,12-15,26,30-36,61H,6-7,10-11,16-25H2,1-2H3,(H2,48,62)(H2,49,63)(H,53,65)(H,54,64)(H,55,70)(H,56,68)(H,57,67)(H,58,66)(H,59,69)(H4,50,51,52)/t30-,31-,32-,33-,34-,35-,36-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 3.10 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from vasopressin V2 receptor in rat kidney membranes |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Oxytocin receptor
(RAT) | BDBM50205313

(CHEMBL265859 | d[Leu4]AVP)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@@H]-1-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccccc2)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6](=O)-[#6]-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C47H67N13O11S2/c1-26(2)20-31-41(66)58-34(23-37(48)62)44(69)59-35(46(71)60-18-7-11-36(60)45(70)55-30(10-6-17-52-47(50)51)40(65)53-24-38(49)63)25-73-72-19-16-39(64)54-32(22-28-12-14-29(61)15-13-28)42(67)57-33(43(68)56-31)21-27-8-4-3-5-9-27/h3-5,8-9,12-15,26,30-36,61H,6-7,10-11,16-25H2,1-2H3,(H2,48,62)(H2,49,63)(H,53,65)(H,54,64)(H,55,70)(H,56,68)(H,57,67)(H,58,66)(H,59,69)(H4,50,51,52)/t30-,31-,32-,33-,34-,35-,36-/m0/s1 | PDB
Reactome pathway
KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 481 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from rat OT receptor expressed in CHO cells |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |
Vasopressin V1a receptor
(RAT) | BDBM50205313

(CHEMBL265859 | d[Leu4]AVP)Show SMILES [#6]-[#6](-[#6])-[#6]-[#6@@H]-1-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccccc2)-[#7]-[#6](=O)-[#6@H](-[#6]-c2ccc(-[#8])cc2)-[#7]-[#6](=O)-[#6]-[#6]-[#16]-[#16]-[#6]-[#6@H](-[#7]-[#6](=O)-[#6@H](-[#6]-[#6](-[#7])=O)-[#7]-[#6]-1=O)-[#6](=O)-[#7]-1-[#6]-[#6]-[#6]-[#6@H]-1-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](\[#7])-[#7])-[#6](=O)-[#7]-[#6]-[#6](-[#7])=O Show InChI InChI=1S/C47H67N13O11S2/c1-26(2)20-31-41(66)58-34(23-37(48)62)44(69)59-35(46(71)60-18-7-11-36(60)45(70)55-30(10-6-17-52-47(50)51)40(65)53-24-38(49)63)25-73-72-19-16-39(64)54-32(22-28-12-14-29(61)15-13-28)42(67)57-33(43(68)56-31)21-27-8-4-3-5-9-27/h3-5,8-9,12-15,26,30-36,61H,6-7,10-11,16-25H2,1-2H3,(H2,48,62)(H2,49,63)(H,53,65)(H,54,64)(H,55,70)(H,56,68)(H,57,67)(H,58,66)(H,59,69)(H4,50,51,52)/t30-,31-,32-,33-,34-,35-,36-/m0/s1 | UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 1.25E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
INSERM
Curated by ChEMBL
| Assay Description
Displacement of [3H]AVP from vasopressin V1a receptor in rat liver membrane |
J Med Chem 50: 835-47 (2007)
Article DOI: 10.1021/jm060928n
BindingDB Entry DOI: 10.7270/Q2G161NH |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data



