

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50218200 (3S,4aR,6aR,6bS,8aS,12aS,14aR,14bR)-8a-(hydroxymethyl)-4,4,6a,6b,11,11,14b-heptamethyl-1,2,3,4,4a,5,6,6a,6b,7,8,8a,9,10,11,12,12a,14,14a,14b-icosahydropicen-3-ol::CHEMBL400074::Erythrodiol::uvaol
SMILES: CC1(C)CC[C@]2(CO)CC[C@]3(C)C(=CC[C@@H]4[C@@]5(C)CC[C@H](O)C(C)(C)[C@@H]5CC[C@@]34C)[C@@H]2C1
InChI Key: InChIKey=PSZDOEIIIJFCFE-OSQDELBUSA-N
Data: 7 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Protein-tyrosine phosphatase 1B (Homo sapiens (Human)) | BDBM50218200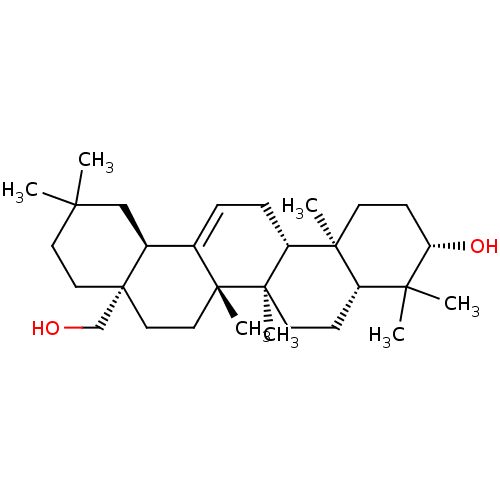 ((3S,4aR,6aR,6bS,8aS,12aS,14aR,14bR)-8a-(hydroxymet...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 9.60E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Universidad Aut£noma del estado de Morelos Curated by ChEMBL | Assay Description Inhibition of purified human GST-tagged PTP-1B using p-nitrophenylphosphate as substrate by spectrophotometry | Eur J Med Chem 87: 316-27 (2014) Article DOI: 10.1016/j.ejmech.2014.09.036 BindingDB Entry DOI: 10.7270/Q2DB83NK | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Protein-tyrosine phosphatase 1B (Homo sapiens (Human)) | BDBM50218200 ((3S,4aR,6aR,6bS,8aS,12aS,14aR,14bR)-8a-(hydroxymet...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >2.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Chinese Academy of Sciences Curated by ChEMBL | Assay Description Inhibition of PTP1B (unknown origin) by pNPP assay | Bioorg Med Chem 16: 8697-705 (2008) Article DOI: 10.1016/j.bmc.2008.07.080 BindingDB Entry DOI: 10.7270/Q2HX1CH7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldo-keto reductase family member 1B10 (AKR1B10) (Homo sapiens (Human)) | BDBM50218200 ((3S,4aR,6aR,6bS,8aS,12aS,14aR,14bR)-8a-(hydroxymet...) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 3.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Gifu Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of reductase activity of N-terminal 6His-tagged human AKR1B10 expressed in Escherichia coli BL21(DE3) assessed as pyridine-3-aldehyde redu... | J Nat Prod 74: 1201-6 (2011) Article DOI: 10.1021/np200118q BindingDB Entry DOI: 10.7270/Q25D8SXZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-lipoxygenase/FLAP (Homo sapiens (Human)) | BDBM50218200 ((3S,4aR,6aR,6bS,8aS,12aS,14aR,14bR)-8a-(hydroxymet...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Osaka University Curated by ChEMBL | Assay Description Inhibition of human recombinant 5-LOX expressed in insect cells assessed as decrease in production of 5-HPETE and 5-HETE using arachidonic acid as su... | J Nat Prod 82: 3311-3320 (2019) Article DOI: 10.1021/acs.jnatprod.9b00538 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nuclear factor NF-kappa-B (Homo sapiens (Human)) | BDBM50218200 ((3S,4aR,6aR,6bS,8aS,12aS,14aR,14bR)-8a-(hydroxymet...) | PDB KEGG UniProtKB/SwissProt B.MOAD antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
The Ohio State University Curated by ChEMBL | Assay Description Inhibition of biotinylated consensus sequence binding to NF-kB p65 in human HeLa nuclear extracts after 3 hrs by ELISA | Bioorg Med Chem 26: 4452-4460 (2018) Article DOI: 10.1016/j.bmc.2018.07.025 BindingDB Entry DOI: 10.7270/Q2N300MM | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-lipoxygenase/FLAP (Homo sapiens (Human)) | BDBM50218200 ((3S,4aR,6aR,6bS,8aS,12aS,14aR,14bR)-8a-(hydroxymet...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | >4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Osaka University Curated by ChEMBL | Assay Description Inhibition of human recombinant 5-LOX expressed in insect cells assessed as decrease in production of 5-HPETE and 5-HETE using arachidonic acid as su... | J Nat Prod 82: 3311-3320 (2019) Article DOI: 10.1021/acs.jnatprod.9b00538 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Aldose reductase (AR) (Homo sapiens (Human)) | BDBM50218200 ((3S,4aR,6aR,6bS,8aS,12aS,14aR,14bR)-8a-(hydroxymet...) | PDB UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | Purchase CHEMBL MCE PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 8.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Gifu Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of reductase activity of N-terminal 6His-tagged human recombinant AKR1B1 expressed in Escherichia coli BL21(DE3) assessed as assessed as p... | J Nat Prod 74: 1201-6 (2011) Article DOI: 10.1021/np200118q BindingDB Entry DOI: 10.7270/Q25D8SXZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||