 Found 14 hits for monomerid = 50236754
Found 14 hits for monomerid = 50236754 Target/Host
(Institution) | Ligand | Target/Host
Links | Ligand
Links | Trg + Lig
Links | Ki
nM | ΔG°
kcal/mole | IC50
nM | Kd
nM | EC50/IC50
nM | koff
s-1 | kon
M-1s-1 | pH | Temp
°C |
|---|
5-hydroxytryptamine receptor 6
(Homo sapiens (Human)) | BDBM50236754
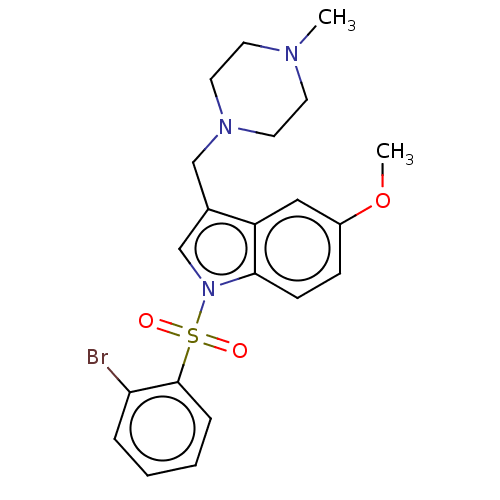
(CHEMBL4082473)Show SMILES O.CS(O)(=O)=O.COc1ccc2n(cc(CN3CCN(C)CC3)c2c1)S(=O)(=O)c1ccccc1Br Show InChI InChI=1S/C21H24BrN3O3S/c1-23-9-11-24(12-10-23)14-16-15-25(20-8-7-17(28-2)13-18(16)20)29(26,27)21-6-4-3-5-19(21)22/h3-8,13,15H,9-12,14H2,1-2H3 | Reactome pathway
KEGG
UniProtKB/SwissProt
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Effective concentration required for partial agonistic activity at Metabotropic glutamate receptor 2; Partial agonist |
J Med Chem 60: 1843-1859 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01662
BindingDB Entry DOI: 10.7270/Q27S7R1X |
More data for this
Ligand-Target Pair | |
D(3) dopamine receptor
(Rattus norvegicus (Rat)) | BDBM50236754

(CHEMBL4082473)Show SMILES O.CS(O)(=O)=O.COc1ccc2n(cc(CN3CCN(C)CC3)c2c1)S(=O)(=O)c1ccccc1Br Show InChI InChI=1S/C21H24BrN3O3S/c1-23-9-11-24(12-10-23)14-16-15-25(20-8-7-17(28-2)13-18(16)20)29(26,27)21-6-4-3-5-19(21)22/h3-8,13,15H,9-12,14H2,1-2H3 | KEGG
UniProtKB/SwissProt
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 616 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Displacement of [3H] 7-OH DPAT from recombinant rat dopamine D3 receptor expressed in CHO cells after 60 mins |
J Med Chem 60: 1843-1859 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01662
BindingDB Entry DOI: 10.7270/Q27S7R1X |
More data for this
Ligand-Target Pair | |
Alpha-2C adrenergic receptor
(Homo sapiens (Human)) | BDBM50236754

(CHEMBL4082473)Show SMILES O.CS(O)(=O)=O.COc1ccc2n(cc(CN3CCN(C)CC3)c2c1)S(=O)(=O)c1ccccc1Br Show InChI InChI=1S/C21H24BrN3O3S/c1-23-9-11-24(12-10-23)14-16-15-25(20-8-7-17(28-2)13-18(16)20)29(26,27)21-6-4-3-5-19(21)22/h3-8,13,15H,9-12,14H2,1-2H3 | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 619 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Inhibitory concentration against Herpes simplex virus type 1 thymidine kinase(HSV-1 TK) |
J Med Chem 60: 1843-1859 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01662
BindingDB Entry DOI: 10.7270/Q27S7R1X |
More data for this
Ligand-Target Pair | |
Alpha-2A adrenergic receptor
(Homo sapiens (Human)) | BDBM50236754

(CHEMBL4082473)Show SMILES O.CS(O)(=O)=O.COc1ccc2n(cc(CN3CCN(C)CC3)c2c1)S(=O)(=O)c1ccccc1Br Show InChI InChI=1S/C21H24BrN3O3S/c1-23-9-11-24(12-10-23)14-16-15-25(20-8-7-17(28-2)13-18(16)20)29(26,27)21-6-4-3-5-19(21)22/h3-8,13,15H,9-12,14H2,1-2H3 | UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| 2.57E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Displacement of [3H] MK-912 from human adrenergic alpha 2A receptor expressed in HT29 cells after 60 mins |
J Med Chem 60: 1843-1859 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01662
BindingDB Entry DOI: 10.7270/Q27S7R1X |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C9
(Homo sapiens (Human)) | BDBM50236754

(CHEMBL4082473)Show SMILES O.CS(O)(=O)=O.COc1ccc2n(cc(CN3CCN(C)CC3)c2c1)S(=O)(=O)c1ccccc1Br Show InChI InChI=1S/C21H24BrN3O3S/c1-23-9-11-24(12-10-23)14-16-15-25(20-8-7-17(28-2)13-18(16)20)29(26,27)21-6-4-3-5-19(21)22/h3-8,13,15H,9-12,14H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C9 in human liver microsomes in presence of NADPH by LC-MS/MS analysis |
J Med Chem 60: 1843-1859 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01662
BindingDB Entry DOI: 10.7270/Q27S7R1X |
More data for this
Ligand-Target Pair | |
Cytochrome P450 1A2
(Homo sapiens (Human)) | BDBM50236754

(CHEMBL4082473)Show SMILES O.CS(O)(=O)=O.COc1ccc2n(cc(CN3CCN(C)CC3)c2c1)S(=O)(=O)c1ccccc1Br Show InChI InChI=1S/C21H24BrN3O3S/c1-23-9-11-24(12-10-23)14-16-15-25(20-8-7-17(28-2)13-18(16)20)29(26,27)21-6-4-3-5-19(21)22/h3-8,13,15H,9-12,14H2,1-2H3 | PDB
UniProtKB/SwissProt
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Inhibition of CYP1A2 in human liver microsomes in presence of NADPH by LC-MS/MS analysis |
J Med Chem 60: 1843-1859 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01662
BindingDB Entry DOI: 10.7270/Q27S7R1X |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50236754

(CHEMBL4082473)Show SMILES O.CS(O)(=O)=O.COc1ccc2n(cc(CN3CCN(C)CC3)c2c1)S(=O)(=O)c1ccccc1Br Show InChI InChI=1S/C21H24BrN3O3S/c1-23-9-11-24(12-10-23)14-16-15-25(20-8-7-17(28-2)13-18(16)20)29(26,27)21-6-4-3-5-19(21)22/h3-8,13,15H,9-12,14H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.50E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Inhibition of CYP3A4 in human liver microsomes using midazolam as substrate in presence of NADPH by LC-MS/MS analysis |
J Med Chem 60: 1843-1859 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01662
BindingDB Entry DOI: 10.7270/Q27S7R1X |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C8
(Homo sapiens (Human)) | BDBM50236754

(CHEMBL4082473)Show SMILES O.CS(O)(=O)=O.COc1ccc2n(cc(CN3CCN(C)CC3)c2c1)S(=O)(=O)c1ccccc1Br Show InChI InChI=1S/C21H24BrN3O3S/c1-23-9-11-24(12-10-23)14-16-15-25(20-8-7-17(28-2)13-18(16)20)29(26,27)21-6-4-3-5-19(21)22/h3-8,13,15H,9-12,14H2,1-2H3 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Inhibition of CYP2C8 in human liver microsomes in presence of NADPH by LC-MS/MS analysis |
J Med Chem 60: 1843-1859 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01662
BindingDB Entry DOI: 10.7270/Q27S7R1X |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2B6
(Homo sapiens (Human)) | BDBM50236754

(CHEMBL4082473)Show SMILES O.CS(O)(=O)=O.COc1ccc2n(cc(CN3CCN(C)CC3)c2c1)S(=O)(=O)c1ccccc1Br Show InChI InChI=1S/C21H24BrN3O3S/c1-23-9-11-24(12-10-23)14-16-15-25(20-8-7-17(28-2)13-18(16)20)29(26,27)21-6-4-3-5-19(21)22/h3-8,13,15H,9-12,14H2,1-2H3 | PDB
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Inhibitory concentration required for antagonistic activity at Metabotropic glutamate receptor 2 |
J Med Chem 60: 1843-1859 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01662
BindingDB Entry DOI: 10.7270/Q27S7R1X |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2D6
(Homo sapiens (Human)) | BDBM50236754

(CHEMBL4082473)Show SMILES O.CS(O)(=O)=O.COc1ccc2n(cc(CN3CCN(C)CC3)c2c1)S(=O)(=O)c1ccccc1Br Show InChI InChI=1S/C21H24BrN3O3S/c1-23-9-11-24(12-10-23)14-16-15-25(20-8-7-17(28-2)13-18(16)20)29(26,27)21-6-4-3-5-19(21)22/h3-8,13,15H,9-12,14H2,1-2H3 | PDB
UniProtKB/SwissProt
UniProtKB/TrEMBL
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 1.32E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Ability to displace [125 I]-MIP-1alpha from C-C chemokine receptor type 5 expressed on CHO cell membranes |
J Med Chem 60: 1843-1859 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01662
BindingDB Entry DOI: 10.7270/Q27S7R1X |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2A6
(Homo sapiens (Human)) | BDBM50236754

(CHEMBL4082473)Show SMILES O.CS(O)(=O)=O.COc1ccc2n(cc(CN3CCN(C)CC3)c2c1)S(=O)(=O)c1ccccc1Br Show InChI InChI=1S/C21H24BrN3O3S/c1-23-9-11-24(12-10-23)14-16-15-25(20-8-7-17(28-2)13-18(16)20)29(26,27)21-6-4-3-5-19(21)22/h3-8,13,15H,9-12,14H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Inhibition of CYP2A6 in human liver microsomes in presence of NADPH by LC-MS/MS analysis |
J Med Chem 60: 1843-1859 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01662
BindingDB Entry DOI: 10.7270/Q27S7R1X |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2E1
(Homo sapiens (Human)) | BDBM50236754

(CHEMBL4082473)Show SMILES O.CS(O)(=O)=O.COc1ccc2n(cc(CN3CCN(C)CC3)c2c1)S(=O)(=O)c1ccccc1Br Show InChI InChI=1S/C21H24BrN3O3S/c1-23-9-11-24(12-10-23)14-16-15-25(20-8-7-17(28-2)13-18(16)20)29(26,27)21-6-4-3-5-19(21)22/h3-8,13,15H,9-12,14H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | >1.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Inhibitory concentration against Herpes simplex virus type 1 thymidine kinase(HSV-1 TK) |
J Med Chem 60: 1843-1859 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01662
BindingDB Entry DOI: 10.7270/Q27S7R1X |
More data for this
Ligand-Target Pair | |
Cytochrome P450 3A4
(Homo sapiens (Human)) | BDBM50236754

(CHEMBL4082473)Show SMILES O.CS(O)(=O)=O.COc1ccc2n(cc(CN3CCN(C)CC3)c2c1)S(=O)(=O)c1ccccc1Br Show InChI InChI=1S/C21H24BrN3O3S/c1-23-9-11-24(12-10-23)14-16-15-25(20-8-7-17(28-2)13-18(16)20)29(26,27)21-6-4-3-5-19(21)22/h3-8,13,15H,9-12,14H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
UniProtKB/TrEMBL
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 2.64E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Inhibitory concentration against Herpes simplex virus type 1 thymidine kinase(HSV-1 TK) |
J Med Chem 60: 1843-1859 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01662
BindingDB Entry DOI: 10.7270/Q27S7R1X |
More data for this
Ligand-Target Pair | |
Cytochrome P450 2C19
(Homo sapiens (Human)) | BDBM50236754

(CHEMBL4082473)Show SMILES O.CS(O)(=O)=O.COc1ccc2n(cc(CN3CCN(C)CC3)c2c1)S(=O)(=O)c1ccccc1Br Show InChI InChI=1S/C21H24BrN3O3S/c1-23-9-11-24(12-10-23)14-16-15-25(20-8-7-17(28-2)13-18(16)20)29(26,27)21-6-4-3-5-19(21)22/h3-8,13,15H,9-12,14H2,1-2H3 | PDB
MMDB
Reactome pathway
KEGG
UniProtKB/SwissProt
B.MOAD
DrugBank
antibodypedia
GoogleScholar
AffyNet 
| CHEMBL
PC cid
PC sid
UniChem
Similars
| Article
PubMed
| n/a | n/a | 5.70E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Suven Life Sciences Ltd
Curated by ChEMBL
| Assay Description
Agonist activity in rat at mGlu2 receptor expressed in HEK293 cells |
J Med Chem 60: 1843-1859 (2017)
Article DOI: 10.1021/acs.jmedchem.6b01662
BindingDB Entry DOI: 10.7270/Q27S7R1X |
More data for this
Ligand-Target Pair | |


 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data