

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50238222 CHEMBL4094591
SMILES: [Br-].COc1ccc2C(=O)C(Cc3cc[n+](Cc4ccccc4C)cc3C(C)=O)Cc2c1
InChI Key: InChIKey=JNQWZIGZXZZYKQ-UHFFFAOYSA-M
Data: 2 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cholinesterase (Equus caballus (Horse)) | BDBM50238222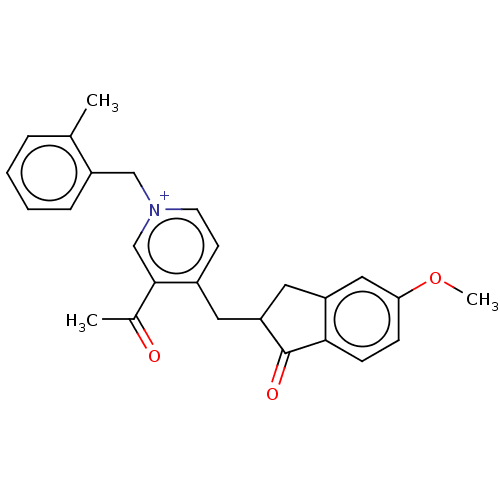 (CHEMBL4094591) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 2.99E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
VFP Therapies Curated by ChEMBL | Assay Description Inhibition of equine serum BChE using butyrylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured every mi... | J Med Chem 60: 5909-5926 (2017) Article DOI: 10.1021/acs.jmedchem.7b00702 BindingDB Entry DOI: 10.7270/Q22V2JC5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Homo sapiens (Human)) | BDBM50238222 (CHEMBL4094591) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | n/a | n/a | 30 | n/a | n/a | n/a | n/a | n/a | n/a |
VFP Therapies Curated by ChEMBL | Assay Description Inhibition of AChE in human erythrocytes using acetylthiocholine iodide as substrate preincubated for 5 mins followed by substrate addition measured ... | J Med Chem 60: 5909-5926 (2017) Article DOI: 10.1021/acs.jmedchem.7b00702 BindingDB Entry DOI: 10.7270/Q22V2JC5 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||