

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50288764 (1S,9R)-13-[2-((1S,2R,5R)-1-Hydroxy-5-isopropyl-2-methyl-cyclohexyl)-2-oxo-acetyl]-3,7-dioxa-13-aza-bicyclo[7.3.1]tridecane-2,8-dione::CHEMBL129672
SMILES: CC(C)[C@@H]1CC[C@@H](C)[C@@](O)(C1)C(=O)C(=O)N1[C@@H]2CCC[C@H]1C(=O)OCCCOC2=O
InChI Key: InChIKey=RCRBJMMTHFNBQP-PXHXRNRRSA-N
Data: 1 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Peptidyl-prolyl cis-trans isomerase FKBP1A (Homo sapiens (Human)) | BDBM50288764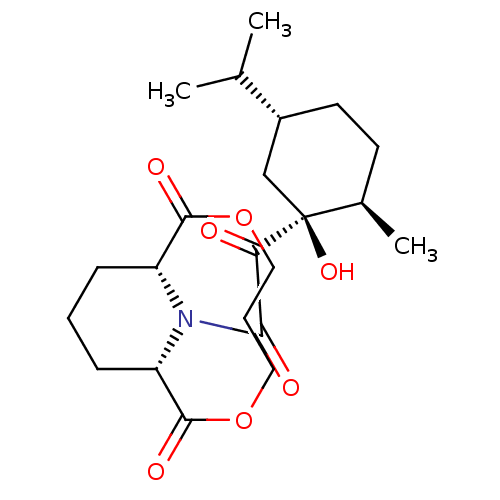 ((1S,9R)-13-[2-((1S,2R,5R)-1-Hydroxy-5-isopropyl-2-...) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article | 8.10E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
TBA Curated by ChEMBL | Assay Description Compound was evaluated for inhibitory activity against human FKBP-12 rotamase | Bioorg Med Chem Lett 6: 385-390 (1996) Article DOI: 10.1016/0960-894X(96)00032-7 BindingDB Entry DOI: 10.7270/Q2862GFJ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||