

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50309861 4-(5-Methyloxazolo[4,5-b]pyridin-2-yl)-1,4-diazabicyclo[3.2.2]-nonane::CHEMBL604798::CP-810123
SMILES: Cc1ccc2oc(nc2n1)N1CCN2CCC1CC2
InChI Key: InChIKey=BSNKYWSMUAGMDO-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Neuronal acetylcholine receptor (Rattus norvegicus (Rat)) | BDBM50309861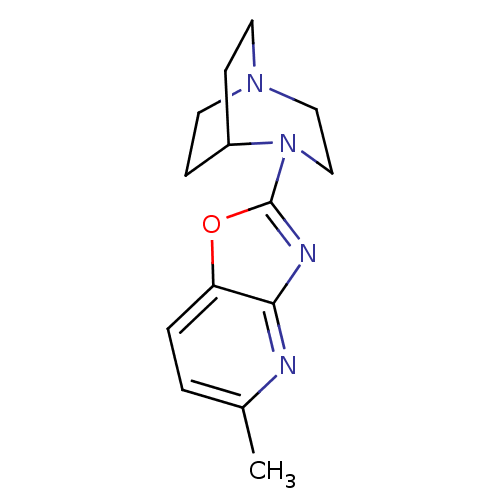 (4-(5-Methyloxazolo[4,5-b]pyridin-2-yl)-1,4-diazabi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 13.5 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development Curated by ChEMBL | Assay Description Displacement of [125I]BTX from rat alpha7 nicotinic acetylcholine receptor expressed in GH4C1 cells | J Med Chem 53: 1222-37 (2010) Article DOI: 10.1021/jm9015075 BindingDB Entry DOI: 10.7270/Q2QN66W3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuronal acetylcholine receptor protein alpha-7 subunit (Homo sapiens (Human)) | BDBM50309861 (4-(5-Methyloxazolo[4,5-b]pyridin-2-yl)-1,4-diazabi...) | PDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 16.4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development Curated by ChEMBL | Assay Description Displacement of [125I]BTX from human alpha7 nicotinic acetylcholine receptor expressed in IMR32 cells | J Med Chem 53: 1222-37 (2010) Article DOI: 10.1021/jm9015075 BindingDB Entry DOI: 10.7270/Q2QN66W3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-HT3A Serotonin Receptor (Mus musculus (house mouse)) | BDBM50309861 (4-(5-Methyloxazolo[4,5-b]pyridin-2-yl)-1,4-diazabi...) | PDB KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 269 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development Curated by ChEMBL | Assay Description Displacement of [3H]LY278584 from mouse 5HT3 receptor expressed in HEK293 cells | J Med Chem 53: 1222-37 (2010) Article DOI: 10.1021/jm9015075 BindingDB Entry DOI: 10.7270/Q2QN66W3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M4 (Homo sapiens (Human)) | BDBM50309861 (4-(5-Methyloxazolo[4,5-b]pyridin-2-yl)-1,4-diazabi...) | Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 3.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development Curated by ChEMBL | Assay Description Inhibition of muscarinic M4 receptor | J Med Chem 53: 1222-37 (2010) Article DOI: 10.1021/jm9015075 BindingDB Entry DOI: 10.7270/Q2QN66W3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M5 (Homo sapiens (Human)) | BDBM50309861 (4-(5-Methyloxazolo[4,5-b]pyridin-2-yl)-1,4-diazabi...) | Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 5.20E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development Curated by ChEMBL | Assay Description Inhibition of muscarinic M5 receptor | J Med Chem 53: 1222-37 (2010) Article DOI: 10.1021/jm9015075 BindingDB Entry DOI: 10.7270/Q2QN66W3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M3 (Homo sapiens (Human)) | BDBM50309861 (4-(5-Methyloxazolo[4,5-b]pyridin-2-yl)-1,4-diazabi...) | Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 8.30E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development Curated by ChEMBL | Assay Description Inhibition of muscarinic M3 receptor | J Med Chem 53: 1222-37 (2010) Article DOI: 10.1021/jm9015075 BindingDB Entry DOI: 10.7270/Q2QN66W3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Muscarinic acetylcholine receptor M1 (Homo sapiens (Human)) | BDBM50309861 (4-(5-Methyloxazolo[4,5-b]pyridin-2-yl)-1,4-diazabi...) | UniProtKB/SwissProt UniProtKB/TrEMBL antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | 1.40E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development Curated by ChEMBL | Assay Description Inhibition of muscarinic M1 receptor | J Med Chem 53: 1222-37 (2010) Article DOI: 10.1021/jm9015075 BindingDB Entry DOI: 10.7270/Q2QN66W3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Potassium voltage-gated channel subfamily H member 2 (Homo sapiens (Human)) | BDBM50309861 (4-(5-Methyloxazolo[4,5-b]pyridin-2-yl)-1,4-diazabi...) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 4.00E+4 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development Curated by ChEMBL | Assay Description Inhibition of human ERG | J Med Chem 53: 1222-37 (2010) Article DOI: 10.1021/jm9015075 BindingDB Entry DOI: 10.7270/Q2QN66W3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| 5-hydroxytryptamine receptor 3A (Homo sapiens (Human)) | BDBM50309861 (4-(5-Methyloxazolo[4,5-b]pyridin-2-yl)-1,4-diazabi...) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Patents Similars | Article PubMed | n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Pfizer Global Research and Development Curated by ChEMBL | Assay Description Agonist activity at human 5HT3 receptor expressed in human skin epithelial cells assessed as stimulation of calcium flux by FLIPR assay | J Med Chem 53: 1222-37 (2010) Article DOI: 10.1021/jm9015075 BindingDB Entry DOI: 10.7270/Q2QN66W3 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||