

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50309995 4-((1H-1,2,4-Triazol-1-yl)methyl)-5-(4-bromophenyl)-N-(1-(4-bromophenyl)ethyl)-1-(2,4-dichlorophenyl)-1H-pyrazole-3-carboxamide::CHEMBL598549
SMILES: Cc1cc(Br)ccc1CNC(=O)c1nn(c(c1Cn1cncn1)-c1ccc(Br)cc1)-c1ccc(Cl)cc1Cl
InChI Key: InChIKey=MFICBBWNFIHARA-UHFFFAOYSA-N
Data: 3 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cannabinoid receptor 1 (Rattus norvegicus (rat)) | BDBM50309995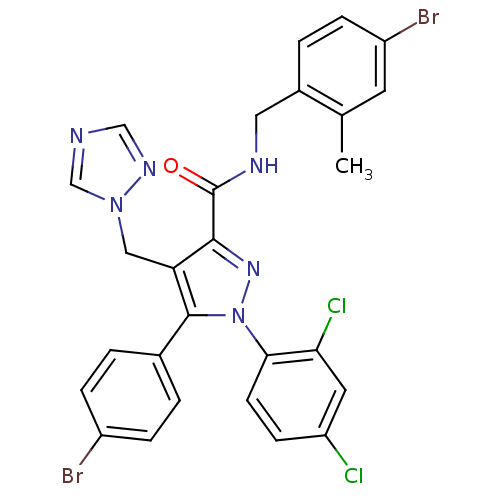 (4-((1H-1,2,4-Triazol-1-yl)methyl)-5-(4-bromophenyl...) | PDB Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 1.40 | n/a | n/a | n/a | n/a | n/a | n/a |
Green Cross Corporation Curated by ChEMBL | Assay Description Displacement of [3H]CP55,940 from CB1 receptor in Sprague-Dawley rat cerebellar membrane | Bioorg Med Chem 18: 1149-62 (2010) Article DOI: 10.1016/j.bmc.2009.12.040 BindingDB Entry DOI: 10.7270/Q2TD9XG2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 1 (Homo sapiens (Human)) | BDBM50309995 (4-((1H-1,2,4-Triazol-1-yl)methyl)-5-(4-bromophenyl...) | PDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 28.2 | n/a | n/a | n/a | n/a | n/a | n/a |
Green Cross Corporation Curated by ChEMBL | Assay Description Inverse agonist activity at human recombinant CB1 receptor expressed in CHO cells by luciferase reporter gene assay | Bioorg Med Chem 18: 1149-62 (2010) Article DOI: 10.1016/j.bmc.2009.12.040 BindingDB Entry DOI: 10.7270/Q2TD9XG2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Cannabinoid receptor 2 (Homo sapiens (Human)) | BDBM50309995 (4-((1H-1,2,4-Triazol-1-yl)methyl)-5-(4-bromophenyl...) | PDB Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 6.48E+3 | n/a | n/a | n/a | n/a | n/a | n/a |
Green Cross Corporation Curated by ChEMBL | Assay Description Displacement of [3H]WIN55,212-2 from human recombinant CB2 receptor expressed in CHO-K1 cell membrane | Bioorg Med Chem 18: 1149-62 (2010) Article DOI: 10.1016/j.bmc.2009.12.040 BindingDB Entry DOI: 10.7270/Q2TD9XG2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||