

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50377431 CHEMBL258744
SMILES: NCCCNc1c2CCCCc2nc2ccccc12
InChI Key: InChIKey=LUDRRADCGGTCHH-UHFFFAOYSA-N
Data: 4 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cholinesterase (Equus caballus (Horse)) | BDBM50377431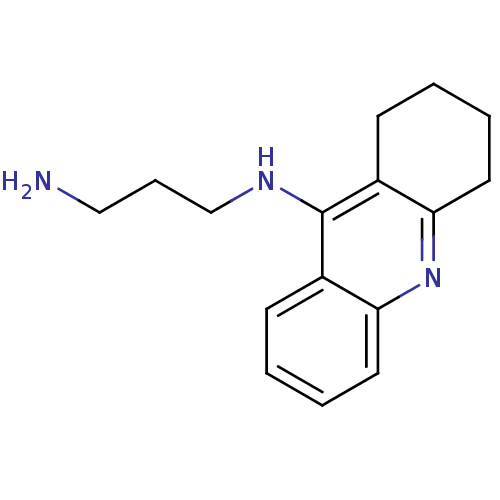 (CHEMBL258744) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 19.7 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of equine serum BuChE using butyrylthiocholine iodide as substrate after 5 mins by Ellman's method | J Med Chem 55: 4309-21 (2012) Article DOI: 10.1021/jm300106z BindingDB Entry DOI: 10.7270/Q21837N7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50377431 (CHEMBL258744) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 69.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Friedrich-Schiller-Universit£t Jena Curated by ChEMBL | Assay Description Inhibition of electric eel AchE by Ellman's assay | Bioorg Med Chem Lett 18: 2905-9 (2008) Article DOI: 10.1016/j.bmcl.2008.03.073 BindingDB Entry DOI: 10.7270/Q2M909KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Carboxylic ester hydrolase (Equus caballus (Horse)) | BDBM50377431 (CHEMBL258744) | PDB MMDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5.5 | n/a | n/a | n/a | n/a | n/a | n/a |
Friedrich-Schiller-Universit£t Jena Curated by ChEMBL | Assay Description Inhibition of equine BChE by Ellman's assay | Bioorg Med Chem Lett 18: 2905-9 (2008) Article DOI: 10.1016/j.bmcl.2008.03.073 BindingDB Entry DOI: 10.7270/Q2M909KN | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholinesterase (Electrophorus electricus (Electric eel)) | BDBM50377431 (CHEMBL258744) | UniProtKB/SwissProt GoogleScholar AffyNet | Purchase CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 79.5 | n/a | n/a | n/a | n/a | n/a | n/a |
China Pharmaceutical University Curated by ChEMBL | Assay Description Inhibition of eel AChE using acetylthiocholine iodide as substrate after 5 mins by Ellman's method | J Med Chem 55: 4309-21 (2012) Article DOI: 10.1021/jm300106z BindingDB Entry DOI: 10.7270/Q21837N7 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||