

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50380752 CHEMBL2017851
SMILES: OC(=O)c1cc(no1)-c1ccc(CC(c2nc(no2)-c2ccccc2)c2ccccc2)cc1
InChI Key: InChIKey=HBPZAUGQOBPKTH-UHFFFAOYSA-N
Data: 1 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Protein-tyrosine phosphatase 1B (Homo sapiens (Human)) | BDBM50380752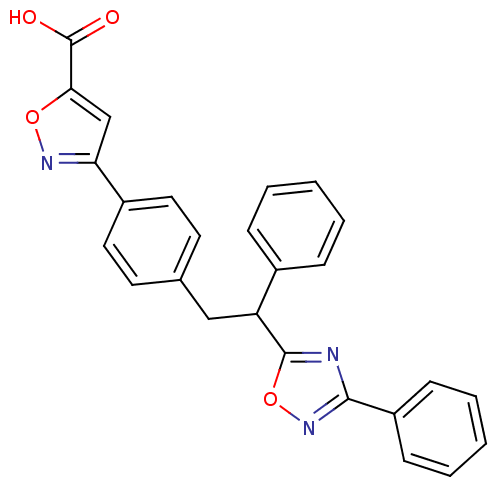 (CHEMBL2017851) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 4.00E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Advinus Therapeutics Ltd Curated by ChEMBL | Assay Description Inhibition of human recombinant PTP1B expressed in Escherichia coli using para-nitrophenyl phosphate as substrate preincubated for 30 mins followed b... | Bioorg Med Chem Lett 22: 2843-9 (2012) Article DOI: 10.1016/j.bmcl.2012.02.070 BindingDB Entry DOI: 10.7270/Q2FB53ZS | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||