

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50382467 CHEMBL2024089
SMILES: CNC(=O)OCC[C@H]1C[C@@H]1c1cncc(OC[C@@H]2CCN2)c1
InChI Key: InChIKey=HGSHFGWWOPXUPH-WHOFXGATSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Acetylcholine Binding protein (Lymnaea stagnalis) | BDBM50382467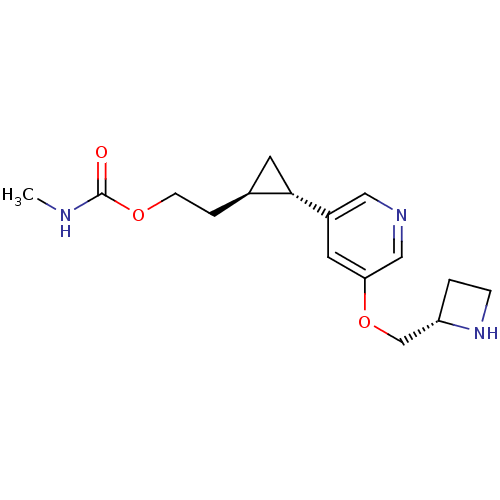 (CHEMBL2024089) | PDB UniProtKB/SwissProt B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 22 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Illinois at Chicago Curated by ChEMBL | Assay Description Displacement of [3H]epibatidine from Lymnaea stagnalis AChBP | J Med Chem 55: 8028-37 (2012) Article DOI: 10.1021/jm3008739 BindingDB Entry DOI: 10.7270/Q2NC62BW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Acetylcholine Binding protein (Aplysia Californica) | BDBM50382467 (CHEMBL2024089) | PDB UniProtKB/TrEMBL B.MOAD GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 1.93E+3 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Illinois at Chicago Curated by ChEMBL | Assay Description Displacement of [3H]epibatidine from Aplysia californica AChBP | J Med Chem 55: 8028-37 (2012) Article DOI: 10.1021/jm3008739 BindingDB Entry DOI: 10.7270/Q2NC62BW | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuronal acetylcholine receptor Alpha-4/Beta-2 (Homo sapiens (Human)) | BDBM50382467 (CHEMBL2024089) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 18.2 | n/a | n/a | n/a | n/a | n/a | n/a |
University of Illinois at Chicago Curated by ChEMBL | Assay Description Antagonist activity at human alpha4beta2 nAChR expressed in SH-EP1 cells assessed as inhibition of carbamylcholine induced 86Rb+ ion efflux preincuba... | J Med Chem 55: 717-24 (2012) Article DOI: 10.1021/jm201157c BindingDB Entry DOI: 10.7270/Q21N8244 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Neuronal acetylcholine receptor Alpha-4/Beta-2 (Homo sapiens (Human)) | BDBM50382467 (CHEMBL2024089) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD DrugBank antibodypedia antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 15.7 | n/a | n/a | n/a | n/a |
University of Illinois at Chicago Curated by ChEMBL | Assay Description Agonist activity at human alpha4beta2 nAChR expressed in SH-EP1 cells assessed as 86Rb+ ion efflux after 9.5 mins by flip-plate technique | J Med Chem 55: 717-24 (2012) Article DOI: 10.1021/jm201157c BindingDB Entry DOI: 10.7270/Q21N8244 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||