

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
null
SMILES: CCN(CC)CCOc1nc(-c2cc(OC)c(Cl)cc2Cl)c2c(c[nH]c2n1)C#N
InChI Key: InChIKey=CPCBFJRWJFEXLH-UHFFFAOYSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Heat shock protein HSP 90-beta (Homo sapiens (Human)) | BDBM50394061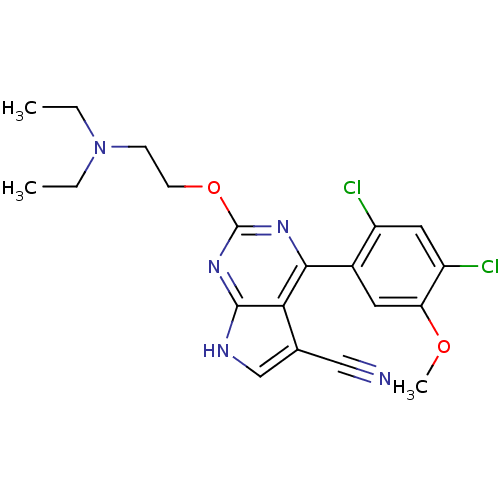 (CHEMBL2158008) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem | Article PubMed | 62 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Vernalis Ltd. Curated by ChEMBL | Assay Description Inhibition of fluorescently labeled VER51001 binding to full length human HSP90beta after 30 mins by fluorescence polarization assay | Bioorg Med Chem 20: 6770-89 (2012) Article DOI: 10.1016/j.bmc.2012.08.050 BindingDB Entry DOI: 10.7270/Q2KS6SN4 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||