

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50399751 CHEMBL2179431
SMILES: [#6]-[#6](-[#6])-[#6@H](-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6](-[#6])-[#6])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]-[#6]-[#7])-[#6](=O)-[#7]-[#6@@H](-[#6]-[#6]-[#6]\[#7]=[#6](/[#7])-[#7])-[#6](-[#8])=O
InChI Key: InChIKey=CSEWNEAENOXTIT-FLMSMKGQSA-N
Data: 2 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Subtilisin/kexin type 6 (Homo sapiens (Human)) | BDBM50399751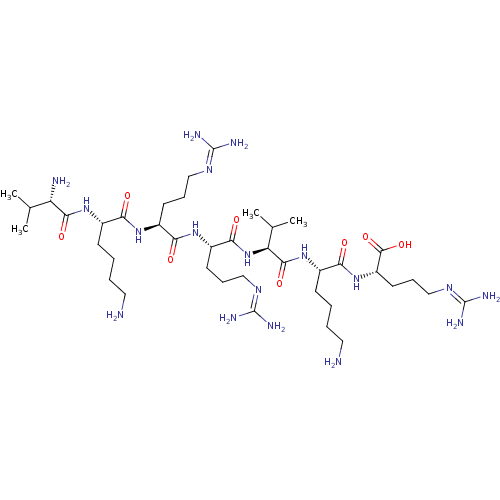 (CHEMBL2179431) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2 | n/a | n/a | n/a | n/a | n/a | n/a | 6.5 | n/a |
Universit£ de Sherbrooke Curated by ChEMBL | Assay Description Competitive inhibition of human recombinant PACE4 expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... | J Med Chem 55: 10501-11 (2012) Article DOI: 10.1021/jm3011178 BindingDB Entry DOI: 10.7270/Q2NV9KDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Homo sapiens furin (paired basic amino acid cleaving enzyme) (FURIN), mRNA (Homo sapiens (Human)) | BDBM50399751 (CHEMBL2179431) | PDB KEGG UniProtKB/SwissProt UniProtKB/TrEMBL B.MOAD antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 5.30 | n/a | n/a | n/a | n/a | n/a | n/a | 7.5 | n/a |
Universit£ de Sherbrooke Curated by ChEMBL | Assay Description Competitive inhibition of human recombinant furin expressed in Drosophila S2 cells using pyroGlu-Arg-Val-Lys-Arg-methyl-coumaryl-7-amide as substrate... | J Med Chem 55: 10501-11 (2012) Article DOI: 10.1021/jm3011178 BindingDB Entry DOI: 10.7270/Q2NV9KDZ | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||