

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50413777 CHEMBL524855
SMILES: CC(C)[C@H](NC(=O)[C@@H]1CSSC(C)(C)[C@@H](NC(=O)[C@@H]2Cc3ccccc3CN2)C(=O)N[C@@H](Cc2ccccc2)C(=O)N[C@@H](Cc2c[nH]c3ccccc23)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](Cc2ccc(O)cc2)C(=O)N1)C(O)=O
InChI Key: InChIKey=GHDRCRPYONKAQM-XJIZABAQSA-N
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Urotensin II receptor (Homo sapiens (Human)) | BDBM50413777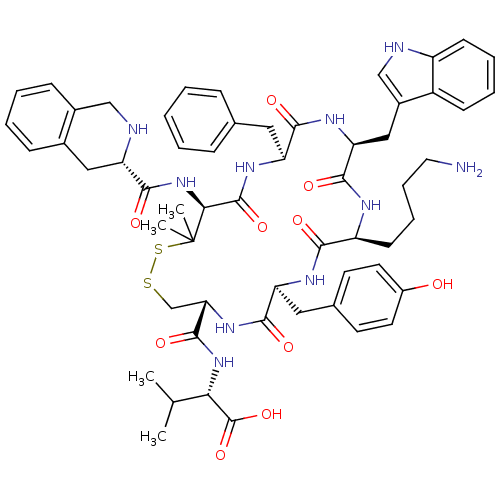 (CHEMBL524855) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 2.63 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
University of Naples Federico II Curated by ChEMBL | Assay Description Displacement of [125I]urotensin-2 from human UT2 receptor expressed in CHO-K1 cells by liquid scintillation counting | J Med Chem 52: 3927-40 (2009) Article DOI: 10.1021/jm900148c BindingDB Entry DOI: 10.7270/Q27W6DFT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| UTS2R (RAT) | BDBM50413777 (CHEMBL524855) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | n/a | n/a | 1.35 | n/a | n/a | n/a | n/a |
University of Naples Federico II Curated by ChEMBL | Assay Description Agonist activity at UT2 receptor in Albino rat aorta assessed as induction of aortic contraction | J Med Chem 52: 3927-40 (2009) Article DOI: 10.1021/jm900148c BindingDB Entry DOI: 10.7270/Q27W6DFT | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||