

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50436959 CHEMBL2402257
SMILES: CCOC(=O)c1cc(C#N)c(nc1C)N1CC(C1)NC(=O)NS(=O)(=O)c1ccccc1
InChI Key: InChIKey=LROWHZKBLPBFIX-UHFFFAOYSA-N
Data: 3 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Purinergic receptor P2Y12 (Homo sapiens (Human)) | BDBM50436959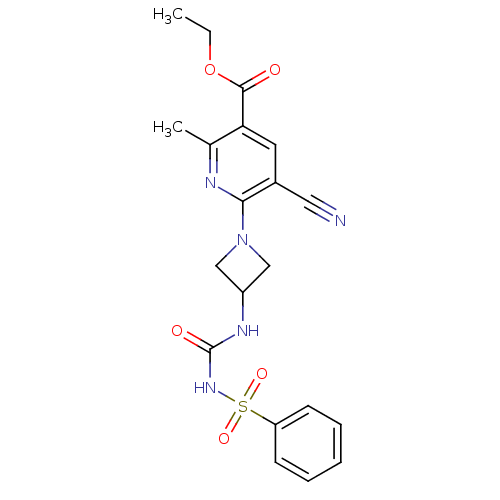 (CHEMBL2402257) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 62 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Curated by ChEMBL | Assay Description Displacement of [125I]-AZ11931285 from P2Y12 receptor (unknown origin) after 1 hr by scintillation counting analysis | Eur J Med Chem 65: 360-75 (2013) Article DOI: 10.1016/j.ejmech.2013.04.007 BindingDB Entry DOI: 10.7270/Q2PK0HJ2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Purinergic receptor P2Y12 (Homo sapiens (Human)) | BDBM50436959 (CHEMBL2402257) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 190 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Curated by ChEMBL | Assay Description Antagonist activity at P2Y12 receptor (unknown origin) assessed as inhibition of GTPgammaS binding after 1 hr by scintillation counting analysis | Eur J Med Chem 65: 360-75 (2013) Article DOI: 10.1016/j.ejmech.2013.04.007 BindingDB Entry DOI: 10.7270/Q2PK0HJ2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Purinergic receptor P2Y12 (Homo sapiens (Human)) | BDBM50436959 (CHEMBL2402257) | PDB Reactome pathway KEGG UniProtKB/SwissProt DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 190 | n/a | n/a | n/a | n/a | n/a | n/a |
AstraZeneca Curated by ChEMBL | Assay Description Antagonist activity at P2Y12 receptor in human washed platelets assessed as inhibition of fibrinogen-induced aggregation | Eur J Med Chem 65: 360-75 (2013) Article DOI: 10.1016/j.ejmech.2013.04.007 BindingDB Entry DOI: 10.7270/Q2PK0HJ2 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||