

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
null
SMILES: Cc1cc(N)nc(COc2cccc(OCC3CCNCC3)c2)c1
InChI Key: InChIKey=PXVODVINMZHSGT-UHFFFAOYSA-N
PDB links: 2 PDB IDs match this monomer.
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Nitric oxide synthase, brain (Homo sapiens (Human)) | BDBM50438642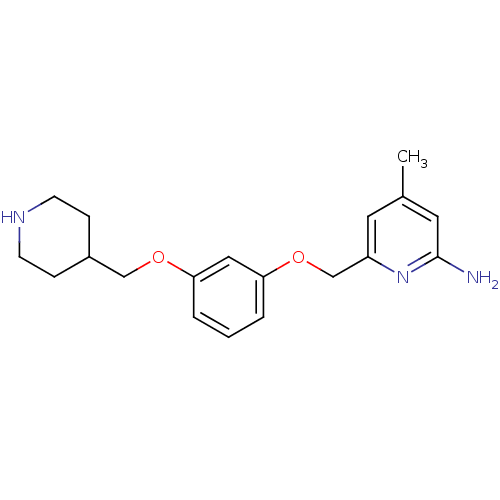 (CHEMBL2414432 | US9732037, Compound 3) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | PDB US Patent | 117 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description NOS inhibition assays of representative compounds 1-21 were undertaken, and the results are summarized in Table 1, below. All NOS isoforms were expre... | US Patent US9732037 (2017) BindingDB Entry DOI: 10.7270/Q26W9D6H | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, brain (Rattus norvegicus (rat)) | BDBM50438642 (CHEMBL2414432 | US9732037, Compound 3) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | PDB Article PubMed | 117 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibition of recombinant rat nNOS expressed in Escherichia coli using L-arginine as substrate assessed as formation of nitric oxide measured up to 6... | Bioorg Med Chem 21: 5323-31 (2013) Article DOI: 10.1016/j.bmc.2013.06.014 BindingDB Entry DOI: 10.7270/Q2PZ5B7N | |||||||||||
| More data for this Ligand-Target Pair |  3D Structure (crystal) | ||||||||||||
| Nitric oxide synthase, inducible (Homo sapiens (Human)) | BDBM50438642 (CHEMBL2414432 | US9732037, Compound 3) | PDB MMDB NCI pathway Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | US Patent | 1.71E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description NOS inhibition assays of representative compounds 1-21 were undertaken, and the results are summarized in Table 1, below. All NOS isoforms were expre... | US Patent US9732037 (2017) BindingDB Entry DOI: 10.7270/Q26W9D6H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, inducible (Mus musculus (mouse)) | BDBM50438642 (CHEMBL2414432 | US9732037, Compound 3) | PDB MMDB Reactome pathway KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 1.71E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibition of recombinant mouse macrophage iNOS expressed in Escherichia coli using L-arginine as substrate assessed as formation of nitric oxide mea... | Bioorg Med Chem 21: 5323-31 (2013) Article DOI: 10.1016/j.bmc.2013.06.014 BindingDB Entry DOI: 10.7270/Q2PZ5B7N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Homo sapiens (Human)) | BDBM50438642 (CHEMBL2414432 | US9732037, Compound 3) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | US Patent | 2.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University US Patent | Assay Description NOS inhibition assays of representative compounds 1-21 were undertaken, and the results are summarized in Table 1, below. All NOS isoforms were expre... | US Patent US9732037 (2017) BindingDB Entry DOI: 10.7270/Q26W9D6H | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| Nitric oxide synthase, endothelial (Bos taurus (bovine)) | BDBM50438642 (CHEMBL2414432 | US9732037, Compound 3) | PDB MMDB KEGG UniProtKB/SwissProt B.MOAD DrugBank GoogleScholar AffyNet | CHEMBL MMDB PC cid PC sid PDB UniChem Similars | Article PubMed | 2.80E+4 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Northwestern University Curated by ChEMBL | Assay Description Inhibition of recombinant bovine eNOS expressed in Escherichia coli using L-arginine as substrate assessed as formation of nitric oxide measured up t... | Bioorg Med Chem 21: 5323-31 (2013) Article DOI: 10.1016/j.bmc.2013.06.014 BindingDB Entry DOI: 10.7270/Q2PZ5B7N | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||