

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50439649 CHEMBL2419811::US9353075, 7
SMILES: CCCCCCC[C@H](C)OC(=O)N[C@@H]1[C@H](C)OC1=O
InChI Key: InChIKey=ROFODRYNJKSNQS-SDDRHHMPSA-N
Data: 4 IC50
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| N-acylethanolamine-hydrolyzing acid amidase (Rattus norvegicus (Rat)) | BDBM50439649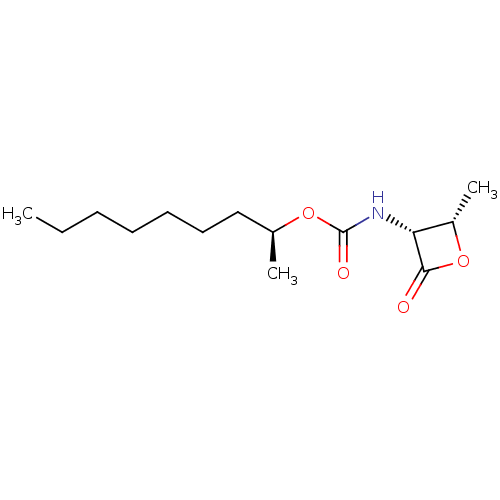 (CHEMBL2419811 | US9353075, 7) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 16 | n/a | n/a | n/a | n/a | 7.4 | n/a |
The Regents of the University of California; Fondazione Istituto Italiano Di Technologia; Universita Degli Studi Di Parma; Universita Degli Studi Di Urbino “Carlo Bo” US Patent | Assay Description Lysosomal NAAA protein preparation were obtained by homogenizing male Sprague-Dawley rat lungs (Charles River) in 20 mM Tris-HCl buffer pH 7.4 contai... | US Patent US9353075 (2016) BindingDB Entry DOI: 10.7270/Q23N229Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| N-acylethanolamine-hydrolyzing acid amidase (Homo sapiens (Human)) | BDBM50439649 (CHEMBL2419811 | US9353075, 7) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 5 | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Inhibition of C-terminal His-6-tagged recombinant human spleen NAAA enzyme expressed in HEK293 cells | J Med Chem 56: 6917-34 (2013) Article DOI: 10.1021/jm400739u BindingDB Entry DOI: 10.7270/Q2F47QK0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| N-acylethanolamine-hydrolyzing acid amidase (Rattus norvegicus (Rat)) | BDBM50439649 (CHEMBL2419811 | US9353075, 7) | PDB UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | n/a | n/a | 16 | n/a | n/a | n/a | n/a | n/a | n/a |
Istituto Italiano di Tecnologia Curated by ChEMBL | Assay Description Inhibition of Sprague Dawley rat lung native NAAA enzyme using heptadecenoylethanolamide as substrate preincubated for 30 mins followed by substrate ... | J Med Chem 56: 6917-34 (2013) Article DOI: 10.1021/jm400739u BindingDB Entry DOI: 10.7270/Q2F47QK0 | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| N-acylethanolamine-hydrolyzing acid amidase (Homo sapiens (Human)) | BDBM50439649 (CHEMBL2419811 | US9353075, 7) | PDB UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | US Patent | n/a | n/a | 5 | n/a | n/a | n/a | n/a | 4.5 | 37 |
The Regents of the University of California; Fondazione Istituto Italiano Di Technologia; Universita Degli Studi Di Parma; Universita Degli Studi Di Urbino “Carlo Bo” US Patent | Assay Description NAAA protein preparation (10 ug) was pre-incubated with various concentrations of test compound or vehicle control in 100 mM NaH2PO4, 100 mM Tri Sodi... | US Patent US9353075 (2016) BindingDB Entry DOI: 10.7270/Q23N229Q | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||