

 Search and Browse
Search and Browse
 Download
Download
 Enter Data
Enter Data
BDBM50442980 CHEMBL3086281
SMILES: CNC(N)=NCCC[C@H](NC(=O)[C@H](CC(C)C)NC(=O)NNC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](CO)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](Cc1ccccc1)NC(=O)[C@H](N)Cc1ccc(O)cc1)C(=O)N[C@@H](Cc1ccccc1)C(N)=O
InChI Key: InChIKey=KRFPQXDNIGUKSZ-BWLQCVBFSA-N
Data: 2 KI
| Target/Host (Institution) | Ligand | Target/Host Links | Ligand Links | Trg + Lig Links | Ki nM | ΔG° kcal/mole | IC50 nM | Kd nM | EC50/IC50 nM | koff s-1 | kon M-1s-1 | pH | Temp °C |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| G-protein Coupled Receptor 54 (Homo sapiens (Human)) | BDBM50442980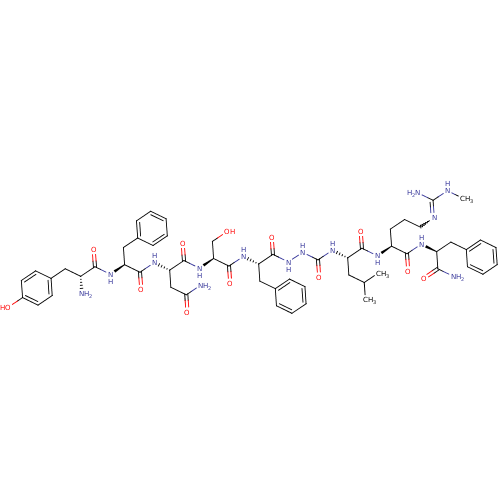 (CHEMBL3086281) | Reactome pathway KEGG UniProtKB/SwissProt antibodypedia GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.0860 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Ltd Curated by ChEMBL | Assay Description Binding affinity to human KISS1R expressed in CHO cell membranes | J Med Chem 56: 8298-307 (2013) Article DOI: 10.1021/jm401056w BindingDB Entry DOI: 10.7270/Q25M675S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||
| KiSS-1 receptor (Rattus norvegicus) | BDBM50442980 (CHEMBL3086281) | Reactome pathway KEGG UniProtKB/SwissProt GoogleScholar AffyNet | CHEMBL PC cid PC sid UniChem Similars | Article PubMed | 0.150 | n/a | n/a | n/a | n/a | n/a | n/a | n/a | n/a |
Takeda Pharmaceutical Company Ltd Curated by ChEMBL | Assay Description Binding affinity to rat KISS1R | J Med Chem 56: 8298-307 (2013) Article DOI: 10.1021/jm401056w BindingDB Entry DOI: 10.7270/Q25M675S | |||||||||||
| More data for this Ligand-Target Pair | |||||||||||||